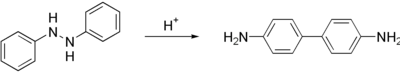Wikiversity
Contents

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
[1,1′-Biphenyl]-4,4′-diamine | |
| Other names
Benzidine, di-phenylamine, diphenylamine, 4,4'-bianiline, 4,4'-biphenyldiamine, 1,1'-biphenyl-4,4'-diamine, 4,4'-diaminobiphenyl, p-diaminodiphenyl, p-benzidine
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.000 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1885 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H12N2 | |
| Molar mass | 184.24 g/mol |
| Appearance | Grayish-yellow, reddish-gray, or white crystalline powder[1] |
| Density | 1.25 g/cm3 |
| Melting point | 122 to 125 °C (252 to 257 °F; 395 to 398 K) |
| Boiling point | 400 °C (752 °F; 673 K) |
| 0.94 g/100 mL at 100 °C | |
| -110.9·10−6 cm3/mol | |
| Related compounds | |
Related compounds
|
biphenyl |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
carcinogenic |
| GHS labelling: | |
  
| |
| Danger | |
| H302, H350, H410 | |
| P201, P202, P264, P270, P273, P281, P301+P312, P308+P313, P330, P391, P405, P501 | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
occupational carcinogen[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Benzidine (trivial name), also called 1,1'-biphenyl-4,4'-diamine (systematic name), is an organic compound with the formula (C6H4NH2)2. It is an aromatic amine. It is a component of a test for cyanide. Related derivatives are used in the production of dyes. Benzidine has been linked to bladder and pancreatic cancer.[2]
Synthesis and properties
Benzidine is prepared in a two step process from nitrobenzene. First, the nitrobenzene is converted to 1,2-diphenylhydrazine, usually using iron powder as the reducing agent. Treatment of this hydrazine with mineral acids induces a rearrangement reaction to 4,4'-benzidine. Smaller amounts of other isomers are also formed.[3] The benzidine rearrangement, which proceeds intramolecularly, is a classic mechanistic puzzle in organic chemistry.[4]
The conversion is described as a [5,5]sigmatropic reaction.[5][6]
In terms of its physical properties, 4,4'-benzidine is poorly soluble in cold water but can be recrystallized from hot water, where it crystallises as the monohydrate. It is dibasic, the deprotonated species has Ka values of 9.3 × 10−10 and 5.6 × 10−11. Its solutions react with oxidizing agents to give deeply coloured quinone-related derivatives.
Applications
Conversion of benzidine to the bis(diazonium) salt was once an integral step in the preparation of direct dyes (requiring no mordant). Treatment of this bis(diazonium) salt with 1-aminonaphthalene-4-sulfonic acid gives the once popular congo red dye. In the past, benzidine was used to test for blood. An enzyme in blood causes the oxidation of benzidine to a distinctively blue-coloured derivative. The test for cyanide relies on similar reactivity. Such applications have largely been replaced by methods using phenolphthalein/hydrogen peroxide and luminol.
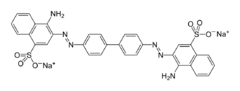
Related 4,4’-benzidines
A variety of derivatives of 4,4’-benzidine are commercially produced on the scale of one to a few thousand kilograms per year, mainly as precursors to dyes and pigments.[3] These derivatives include, in order of scale, the following:
- 3,3'-Dichlorobenzidine
- o-tolidine, 3,3'-dimethyl-4,4’-benzidine
- o-dianisidine (3,3'-dimethoxy-4,4’-benzidine, CAS# 119-90-4, m.p. 133 °C)
- 3,3',4,4'-Tetraamino-diphenyl, precursor to polybenzimidazole fiber.
Safety
As with some other aromatic amines such as 2-naphthylamine, benzidine has been significantly withdrawn from use in most industries because it is so carcinogenic. In August 2010 benzidine dyes were included in the U.S. EPA's List of Chemicals of Concern.[7] The manufacture of Benzidine has been illegal in the UK since at least 2002 under the Control of Substances Hazardous to Health Regulations 2002 (COSHH).
References
- ^ a b NIOSH Pocket Guide to Chemical Hazards. "#0051". National Institute for Occupational Safety and Health (NIOSH).
- ^ "Known and Probable Carcinogens". American Cancer Society. 2011-06-29. Archived from the original on 2008-03-17. Retrieved 2007-01-12.
- ^ a b Schwenecke, H.; Mayer, D. (2005). "Benzidine and Benzidine Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_539. ISBN 978-3527306732.
- ^ March, J. (1992). Advanced Organic Chemistry (5th ed.). New York: J. Wiley and Sons. ISBN 0-471-60180-2.
- ^ Shine, H. J.; Zmuda, H.; Park, K. H.; Kwart, H.; Horgan, A. G.; Collins, C.; Maxwell, B. E. (1981). "Mechanism of the benzidine rearrangement. Kinetic isotope effects and transition states. Evidence for concerted rearrangement". Journal of the American Chemical Society. 103 (4): 955–956. doi:10.1021/ja00394a047..
- ^ Shine, H. J.; Zmuda, H.; Park, K. H.; Kwart, H.; Horgan, A. G.; Brechbiel, M. (1982). "Benzidine rearrangements. 16. The use of heavy-atom kinetic isotope effects in solving the mechanism of the acid-catalyzed rearrangement of hydrazobenzene. The concerted pathway to benzidine and the nonconcerted pathway to diphenyline". Journal of the American Chemical Society. 104 (9): 2501–2509. doi:10.1021/ja00373a028.
- ^ "Benzidine Dyes Action Plan Summary". U. S. Environmental Protection Agency. 2010-08-18. Archived from the original on 2010-08-21.