LIMSwiki
Contents
 | |
| Clinical data | |
|---|---|
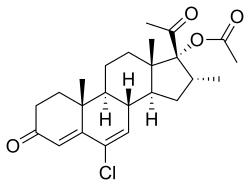
| Other names | Clomagestone acetate; SH-741; 6-Chloro-17α-acetoxy-16α-methylpregna-4,6-diene-3,20-dione |
| Drug class | Progestogen; Progestogen ester |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C24H31ClO4 |
| Molar mass | 418.96 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Clomegestone acetate (USAN) (developmental code name SH-741), or clomagestone acetate, also known as 6-chloro-17α-acetoxy-16α-methylpregna-4,6-diene-3,20-dione, is a steroidal progestin of the 17α-hydroxyprogesterone group which was developed as an oral contraceptive but was never marketed.[1][2] It is the acetate ester of clomegestone, which, similarly to clomegestone acetate, was never marketed.[1] Clomegestone acetate is also the 17-desoxy cogener of clometherone, and is somewhat more potent in comparison.[3] Similarly to cyproterone acetate, clomegestone acetate has been found to alter insulin receptor concentrations in adipose tissue,[4] and this may indicate the presence of glucocorticoid activity.[5][6]
References
- ^ a b Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 297–. ISBN 978-1-4757-2085-3.
- ^ Lawrence MM (1971). CENTO Workshop Series on Clinical and Applied Research in Family Planning, Ankara, Turkey, February 23-26: Tehran, Iran, March 1-4; Lahore, Pakistan, March 9-12, 1971; [papers. Central Treaty Organization.
- ^ Lednicer D (20 June 2011). Steroid Chemistry at a Glance. John Wiley & Sons. pp. 5–. ISBN 978-1-119-95729-4.
- ^ Krauth MC, Schillinger E (November 1977). "Changes in insulin receptor concentration in rat fat cells following treatment with the gestagens clomegestone acetate and cyproterone acetate". Acta Endocrinologica. 86 (3): 667–672. doi:10.1530/acta.0.0860667. PMID 579029.
- ^ Krauth MC, Schillinger E (November 1977). "Changes in insulin receptor concentration in rat fat cells following treatment with the gestagens clomegestone acetate and cyproterone acetate". Acta Endocrinologica. 86 (3): 667–672. doi:10.1530/acta.0.0860667. PMID 579029.
- ^ Schillinger E, Gerloff C, Gerhards E, Günzel P (February 1974). "Glucose tolerance and serum insulin in rats and insulin sensitivity of rat adipose cells following treatment with the progestogen clomegestone acetate". Acta Endocrinologica. 75 (2): 305–313. doi:10.1530/acta.0.0750305. PMID 4406547.

















