Trends in LIMS
Contents

In organic chemistry, nitroso refers to a functional group in which the nitric oxide (−N=O) group is attached to an organic moiety. As such, various nitroso groups can be categorized as C-nitroso compounds (e.g., nitrosoalkanes; R−N=O), S-nitroso compounds (nitrosothiols; RS−N=O), N-nitroso compounds (e.g., nitrosamines, RN(−R’)−N=O), and O-nitroso compounds (alkyl nitrites; RO−N=O).
Synthesis
Nitroso compounds can be prepared by the reduction of nitro compounds[1] or by the oxidation of hydroxylamines.[2] Ortho-nitrosophenols may be produced by the Baudisch reaction. In the Fischer–Hepp rearrangement, aromatic 4-nitrosoanilines are prepared from the corresponding nitrosamines.
Properties
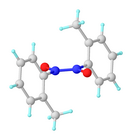
Nitrosoarenes typically participate in a monomer–dimer equilibrium. The azobenzene N,N'-dioxide (Ar(–O)N+=+N(O–)Ar) dimers, which are often pale yellow, are generally favored in the solid state, whereas the deep-green monomers are favored in dilute solution or at higher temperatures. They exist as cis and trans isomers.[4] The central "double bond" in the dimer in fact has a bond order of about 1.5.[5]
When stored in protic media, primary and secondary nitrosoalkanes isomerize to oximes.[6] Some tertiary nitrosoalkanes also isomerize to oximes through C-C bond fission, particularly if the bond is electron-poor.[7] Nitrosophenols and naphthols isomerize to the oxime quinone in solution, but reversibly; nitrosophenol ethers typically dealkylate to facilitate the isomerization. Nitroso tertiary anilines generally do not dealkylate in that way.[8]
Due to the stability of the nitric oxide free radical, nitroso organyls tend to have very low C–N bond dissociation energies: nitrosoalkanes have BDEs on the order of 30–40 kcal/mol (130–170 kJ/mol), while nitrosoarenes have BDEs on the order of 50–60 kcal/mol (210–250 kJ/mol). As a consequence, they are generally heat- and light-sensitive. Compounds containing O–(NO) or N–(NO) bonds generally have even lower bond dissociation energies. For instance, N-nitrosodiphenylamine, Ph2N–N=O, has a N–N bond dissociation energy of only 23 kcal/mol (96 kJ/mol).[9]
Organonitroso compounds serve as a ligands giving transition metal nitroso complexes.[10]
Reactions
Many reactions make use of an intermediate nitroso compound, such as the Barton reaction and Davis–Beirut reaction, as well as the synthesis of indoles, for example: Baeyer–Emmerling indole synthesis, Bartoli indole synthesis. In the Saville reaction, mercury is used to replace a nitrosyl from a thiol group.
C-nitroso compounds are used in organic synthesis as synthons in some well-documented chemical reactions such as hetero Diels-Alder (HDA), nitroso-ene and nitroso-aldol reactions.[11]
Nitrosyl in inorganic chemistry

Nitrosyls are non-organic compounds containing the NO group, for example directly bound to the metal via the N atom, giving a metal–NO moiety. Alternatively, a nonmetal example is the common reagent nitrosyl chloride (Cl−N=O). Nitric oxide is a stable radical, having an unpaired electron. Reduction of nitric oxide gives the nitrosyl anion, NO−:
- NO + e− → NO−
Oxidation of NO yields the nitrosonium cation, NO+:
- NO → NO+ + e−
Nitric oxide can serve as a ligand forming metal nitrosyl complexes or just metal nitrosyls. These complexes can be viewed as adducts of NO+, NO−, or some intermediate case.
In human health
Nitroso compounds react with primary amines in acidic environments to form nitrosamines, which human metabolism converts to mutagenic diazo compounds. Small amounts of nitro and nitroso compounds form during meat curing; the toxicity of these compounds preserves the meat against bacterial infection. After curing completes, the concentration of these compounds appears to degrade over time. Their presence in finished products has been tightly regulated since several food-poisoning cases in the early 20th century,[12] but consumption of large quantities of processed meats can still cause a slight elevation in gastric and oesophageal cancer risk today.[13][14][15][16]
For example, during the 1970s, certain Norwegian farm animals began exhibiting elevated levels of liver cancer. These animals had been fed herring meal preserved with sodium nitrite. The sodium nitrite had reacted with dimethylamine in the fish and produced dimethylnitrosamine.[17]
The effects of nitroso compounds vary dramatically across the gastrointestinal tract, and with diet. Nitroso compounds present in stool do not induce nitrosamine formation, because stool has neutral pH.[18][19] Stomach acid does cause nitrosamine compound formation, but the process is inhibited when amine concentration is low (e.g. a low-protein diet or no fermented food). The process may also be inhibited in the case of high vitamin C (ascorbic acid) concentration (e.g. high-fruit diet).[20][21][22] However, when 10% of the meal is fat, the effect reverses, and ascorbic acid markedly increases nitrosamine formation.[23][24]
See also
- Nitrosamine, the functional group with the NO attached to an amine, such as R2N–NO
- Nitrosobenzene
- Nitric oxide
- Nitroxyl
References
- ^ G. H. Coleman; C. M. McCloskey; F. A. Stuart (1945). "Nitrosobenzene". Org. Synth. 25: 80. doi:10.15227/orgsyn.025.0080.
- ^ Calder, A.; Forrester, A. R.; Hepburn, S. P. "2-Methyl-2-nitrosopropane and Its Dimer". Organic Syntheses. 52: 77; Collected Volumes, vol. 6, p. 803.
- ^ E.Bosch (2014). "Structural Analysis of Methyl-Substituted Nitrosobenzenes and Nitrosoanisoles". J. Chem. Cryst. 98 (2): 44. doi:10.1007/s10870-013-0489-8. S2CID 95291018.
- ^ Beaudoin, D.; Wuest, J. D. (2016). "Dimerization of Aromatic C-Nitroso Compounds". Chemical Reviews. 116 (1): 258–286. doi:10.1021/cr500520s. PMID 26730505.
- ^ Williams, D. L. H. (1988). Nitrosation. Cambridge, UK: Cambridge University. p. 36. ISBN 0-521-26796-X.
- ^ Kirby, G. W. (1977). "Electrophilic C-nitroso-compounds". Chemical Society Reviews. 6: 2. doi:10.1039/CS9770600001 (Tilden lecture).
- ^ Williams 1988, p. 36.
- ^ Williams 1988, pp. 59–61.
- ^ Luo, Yu-Ran (2007). Comprehensive Handbook of Chemical Bond Energies. Boca Raton, FL: Taylor and Francis. ISBN 9781420007282.
- ^ Lee, Jonghyuk; Chen, Li; West, Ann H.; Richter-Addo, George B. (2002). "Interactions of Organic Nitroso Compounds with Metals". Chemical Reviews. 102 (4): 1019–1066. doi:10.1021/cr0000731. PMID 11942786.
- ^ Bianchi, P.; Monbaliu, J. C. M. (2022). "Three decades of unveiling the complex chemistry of C-nitroso species with computational chemistry". Organic Chemistry Frontiers. 9: 223–264. doi:10.1039/d1qo01415c.
- ^ Honikel, K. O. (2008). "The use an control of nitrate and nitrite for the processing of meat products" (PDF). Meat Science. 78 (1–2): 68–76. doi:10.1016/j.meatsci.2007.05.030. PMID 22062097.
- ^ Lunn, J.C.; Kuhnle, G.; Mai, V.; Frankenfeld, C.; Shuker, D.E.G.; Glen, R. C.; Goodman, J.M.; Pollock, J.R.A.; Bingham, S.A. (2006). "The effect of haem in red and processed meat on the endogenous formation of N-nitroso compounds in the upper gastrointestinal tract". Carcinogenesis. 28 (3): 685–690. doi:10.1093/carcin/bgl192. PMID 17052997.
- ^ Bastide, Nadia M.; Pierre, Fabrice H.F.; Corpet, Denis E. (2011). "Heme Iron from Meat and Risk of Colorectal Cancer: A Meta-analysis and a Review of the Mechanisms Involved". Cancer Prevention Research. 4 (2): 177–184. doi:10.1158/1940-6207.CAPR-10-0113. PMID 21209396. S2CID 4951579.
- ^ Bastide, Nadia M.; Chenni, Fatima; Audebert, Marc; Santarelli, Raphaelle L.; Taché, Sylviane; Naud, Nathalie; Baradat, Maryse; Jouanin, Isabelle; Surya, Reggie; Hobbs, Ditte A.; Kuhnle, Gunter G.; Raymond-Letron, Isabelle; Gueraud, Françoise; Corpet, Denis E.; Pierre, Fabrice H.F. (2015). "A Central Role for Heme Iron in Colon Carcinogenesis Associated with Red Meat Intake". Cancer Research. 75 (5): 870–879. doi:10.1158/0008-5472.CAN-14-2554. PMID 25592152. S2CID 13274953.
- ^ Jakszyn, P; Gonzalez, CA (2006). "Nitrosamine and related food intake and gastric and oesophageal cancer risk: A systematic review of the epidemiological evidence". World Journal of Gastroenterology. 12 (27): 4296–4303. doi:10.3748/wjg.v12.i27.4296. PMC 4087738. PMID 16865769.
- ^ Joyce I. Boye; Yves Arcand (2012-01-10). Green Technologies in Food Production and Processing. Springer Science & Business Media. p. 573. ISBN 978-1-4614-1586-2.
- ^ Lee, L; Archer, MC; Bruce, WR (October 1981). "Absence of volatile nitrosamines in human feces". Cancer Res. 41 (10): 3992–4. PMID 7285009.
- ^ Kuhnle, GG; Story, GW; Reda, T; et al. (October 2007). "Diet-induced endogenous formation of nitroso compounds in the GI tract". Free Radic. Biol. Med. 43 (7): 1040–7. doi:10.1016/j.freeradbiomed.2007.03.011. PMID 17761300.
- ^ Mirvish, SS; Wallcave, L; Eagen, M; Shubik, P (July 1972). "Ascorbate–nitrite reaction: possible means of blocking the formation of carcinogenic N-nitroso compounds". Science. 177 (4043): 65–8. Bibcode:1972Sci...177...65M. doi:10.1126/science.177.4043.65. PMID 5041776. S2CID 26275960.
- ^ Mirvish, SS (October 1986). "Effects of vitamins C and E on N-nitroso compound formation, carcinogenesis, and cancer". Cancer. 58 (8 Suppl): 1842–50. doi:10.1002/1097-0142(19861015)58:8+<1842::aid-cncr2820581410>3.0.co;2-#. PMID 3756808. S2CID 196379002.
- ^ Tannenbaum SR, Wishnok JS, Leaf CD (1991). "Inhibition of nitrosamine formation by ascorbic acid". The American Journal of Clinical Nutrition. 53 (1 Suppl): 247S–250S. Bibcode:1987NYASA.498..354T. doi:10.1111/j.1749-6632.1987.tb23774.x. PMID 1985394. S2CID 41045030. Retrieved 2015-06-06.
Evidence now exists that ascorbic acid is a limiting factor in nitrosation reactions in people.
- ^ Combet, E.; Paterson, S; Iijima, K; Winter, J; Mullen, W; Crozier, A; Preston, T; McColl, K. E. (2007). "Fat transforms ascorbic acid from inhibiting to promoting acid-catalysed N-nitrosation". Gut. 56 (12): 1678–1684. doi:10.1136/gut.2007.128587. PMC 2095705. PMID 17785370.
- ^ Combet, E; El Mesmari, A; Preston, T; Crozier, A; McColl, K. E. (2010). "Dietary phenolic acids and ascorbic acid: Influence on acid-catalyzed nitrosative chemistry in the presence and absence of lipids". Free Radical Biology and Medicine. 48 (6): 763–771. doi:10.1016/j.freeradbiomed.2009.12.011. PMID 20026204.

















