Trends in LIMS
Contents

In chemistry, azide (/ˈeɪzaɪd/, AY-zyd) is a linear, polyatomic anion with the formula N−3 and structure −N=N+=N−. It is the conjugate base of hydrazoic acid HN3. Organic azides are organic compounds with the formula RN3, containing the azide functional group.[1] The dominant application of azides is as a propellant in air bags.[1]
Preparation
Sodium azide is made industrially by the reaction of nitrous oxide, N2O with sodium amide NaNH2 in liquid ammonia as solvent:[2]
Many inorganic azides can be prepared directly or indirectly from sodium azide. For example, lead azide, used in detonators, may be prepared from the metathesis reaction between lead nitrate and sodium azide. An alternative route is direct reaction of the metal with silver azide dissolved in liquid ammonia.[3] Some azides are produced by treating the carbonate salts with hydrazoic acid.
Bonding
Azide is isoelectronic with carbon dioxide CO2, cyanate OCN−, nitrous oxide N2O, nitronium ion NO+2, molecular beryllium fluoride BeF2 and cyanogen fluoride FCN. Per valence bond theory, azide can be described by several resonance structures; an important one being N−=N+=N−.
Reactions
Azide salts can decompose with release of nitrogen gas. The decomposition temperatures of the alkali metal azides are: NaN3 (275 °C), KN3 (355 °C), RbN3 (395 °C), and CsN3 (390 °C). This method is used to produce ultrapure alkali metals:[4]
- 2 MN3 2 M + 3 N2
Protonation of azide salts gives toxic hydrazoic acid in the presence of strong acids:
- H+ + N−3 → HN3
Azide as a ligand forms numerous transition metal azide complexes. Some such compounds are shock sensitive.
Many inorganic covalent azides (e.g., fluorine azide, chlorine azide, bromine azide, iodine azide, silicon tetraazide) have been described.[5]
The azide anion behaves as a nucleophile; it undergoes nucleophilic substitution for both aliphatic and aromatic systems. It reacts with epoxides, causing a ring-opening; it undergoes Michael-like conjugate addition to 1,4-unsaturated carbonyl compounds.[1]
Azides can be used as precursors of the metal nitrido complexes by being induced to release N2, generating a metal complex in unusual oxidation states (see high-valent iron).
Redox behaviour and trend to disproportionation
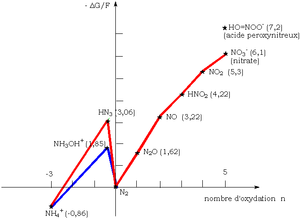
Azides have an ambivalent redox behavior: they are both oxidizing and reducing, as they are easily subject to disproportionation, as illustrated by the Frost diagram of nitrogen. This diagram shows the significant energetic instability of the hydrazoic acid HN3 (or the azide ion) surrounded by two much more stable species, the ammonium ion NH+4 on the left and the molecular nitrogen N2 on the right. As seen on the Frost diagram the disproportionation reaction lowers ∆G, the Gibbs free energy of the system (-∆G/F = zE, where F is the Faraday constant, z the number of electrons exchanged in the redox reaction, and E the standard electrode potential). By minimizing the energy in the system, the disproportionation reaction increases its thermodynamical stability.
Destruction by oxidation by nitrite
Azides decompose with nitrite compounds such as sodium nitrite. Each elementary redox reaction is also a comproportionation reaction because two different N-species (N−3 and NO−2) converge to a same one (respectively N2, N2O and NO) and is favored when the solution is acidified. This is a method of destroying residual azides, prior to disposal.[6] In the process, nitrogen gas (N2) and nitrogen oxides (N2O and NO) are formed:
- 3 N−3 + NO−2 + 2 H2O → 5 N2 + 4 OH−
- 2 N−3 + 4 NO−2 + 3 H2O → 5 N2O + 6 OH−
- N−3 + 7 NO−2 + 4 H2O → 10 NO + 8 OH−
Azide (-⅓) (the reductant, electron donor) is oxidized in N2 (0), nitrous oxide (N2O) (+1), or nitric oxide (NO) (+2) while nitrite (+3) (the oxidant, electron acceptor) is simultaneously reduced to the same corresponding species in each elementary redox reaction considered here above. The respective stability of the reaction products of these three comproportionation redox reactions is in the following order: N2 > N2O > NO, as can be verified in the Frost diagram for nitrogen.
Applications
In 2005, about 251 tons of azide-containing compounds were annually produced in the world, the main product being sodium azide.[7]
Primary explosives and propellants
Sodium azide NaN3 is the propellant in automobile airbags. It decomposes on heating to give nitrogen gas, which is used to quickly expand the air bag:[7]
- 2 NaN3 → 2 Na + 3 N2
Heavy metal azides, such as lead azide, Pb(N3)2, are shock-sensitive detonators which violently decompose to the corresponding metal and nitrogen, for example:[8]
- Pb(N3)2 → Pb + 3 N2
Silver azide AgN3 and barium azide Ba(N3)2 are used similarly.
Some organic azides are potential rocket propellants, an example being 2-dimethylaminoethylazide (DMAZ) (CH3)2NCH2CH2N3.
Microbial inhibitor and undesirable side effects
Sodium azide is commonly used in the laboratory as a bacteriostatic agent to avoid microbial proliferation in abiotic control experiments in which it is important to avoid microbial activity. However, it has the disadvantage to be prone to trigger unexpected and undesirable side reactions that can jeopardize the experimental results. Indeed, the azide anion is a nucleophile and a redox-active species. Being prone to disproportionation, it can behave both as an oxidizing and as a reducing agent. Therefore, it is susceptible to interfere in an unpredictable way with many substances.[9][10][11] For example, the azide anion can oxidize pyrite (FeS2) with the formation of thiosulfate (S2O2−3), or reduce quinone into hydroquinone.[12] It can also reduce nitrite NO−2 into nitrous oxide N2O, and Fe2+ into Fe0 (zerovalent iron, ZVI).[12] Azide can also enhance the N2O emission in soil. A proposed explanation is the stimulation of the denitrification processes because of the azide’s role in the synthesis of denitrifying enzymes.[13] Moreover, azide also affects the absorbance and fluorescence optical properties of the dissolved organic matter (DOM) from soils.[14] Many other interferences are reported in the literature for biochemical and biological analyses and they should be systematically identified and first rigorously tested in the laboratory before to use azide as microbial inhibitor for a given application.
Purification of molten sodium
Sodium azide NaN3 is used to purify metallic sodium in laboratories handling molten sodium used as a coolant for fast-neutron reactors.[15]
As hydrazoic acid, the protonated form of the azide anion, has a very low reduction potential E°red = -3,09 volt, and is even a stronger reductant than lithium (E°red = -3.04 volt), dry solid sodium azide can be added to molten metallic sodium (E°red = -2,71 volt) under strict anoxic conditions (e.g., in a special anaerobic glovebox with very low residual O2 (< 1 ppm vol.) to reduce Na+ impurities still present into the sodium bath. The reaction residue is only gaseous N2.
As E°ox = -E°red, it gives the following series of oxidation reactions when the redox couples are presented as reductants:
- 2 HN3 ⇌ 3 N2(g) + 2 H+ + 2 e− (E°ox = +3,09 volt)
- Li ⇌ Li+ + e− (E°ox = +3,04 volt)
- Na ⇌ Na+ + e− (E°ox = +2,71 volt)
Click chemistry
The azide functional group is commonly utilized in click chemistry through copper(I)-catalyzed azide-alkyne cycloaddition (CuAAC) reactions, where copper(I) catalyzes the cycloaddition of an organoazide to a terminal alkyne, forming a triazole.[16][17][18]
Other uses
A very damaging and illegal usage of sodium azide is its diversion by poachers as a substitute of sodium cyanide to poison some animal species by blocking the electron transport chain in the cellular respiration process.
Safety
Azides are explosophores[9][19][20] and respiratory poisons.[9][21] Sodium azide (NaN3) is as toxic as sodium cyanide (NaCN) (with an oral LD50 of 27 mg/kg in rats) and can be absorbed through the skin. When sodium azide enters in contact with an acid, it produces volatile hydrazoic acid (HN3), as toxic and volatile as hydrogen cyanide (HCN). When accidentally present in the air of a laboratory at low concentration, it can cause irritations such as nasal stuffiness, or suffocation and death at elevated concentrations.[22]
Heavy metal azides, such as lead azide (Pb(N3)2) are primary high explosives detonable when heated or shaken. Heavy-metal azides are formed when solutions of sodium azide or HN3 vapors come into contact with heavy metals (Pb, Hg…) or their salts. Heavy-metal azides can accumulate under certain circumstances, for example, in metal pipelines and on the metal components of diverse equipment (rotary evaporators, freezedrying equipment, cooling traps, water baths, waste pipes), and thus lead to violent explosions.[9]
See also
- Homoleptic azido compounds
- Pentazenium
- Pentazolate (cyclo-N−5)
References
- ^ a b c S. Bräse; C. Gil; K. Knepper; V. Zimmermann (2005). "Organic Azides: An Exploding Diversity of a Unique Class of Compounds". Angewandte Chemie International Edition. 44 (33): 5188–5240. doi:10.1002/anie.200400657. PMID 16100733.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 433. ISBN 978-0-08-037941-8.
- ^ Müller, Thomas G.; Karau, Friedrich; Schnick, Wolfgang; Kraus, Florian (2014). "A New Route to Metal Azides". Angewandte Chemie. 53 (50): 13695–13697. doi:10.1002/anie.201404561. PMID 24924913.
- ^ Dönges, E. (1963). "Alkali Metals". In Brauer, G. (ed.). Handbook of Preparative Inorganic Chemistry. Vol. 1 (2nd ed.). NY: Academic Press. p. 475.
- ^ I. C. Tornieporth-Oetting & T. M. Klapötke (1995). "Covalent Inorganic Azides". Angewandte Chemie International Edition in English. 34 (5): 511–520. doi:10.1002/anie.199505111.
- ^ Committee on Prudent Practices for Handling, Storage, and Disposal of Chemicals in Laboratories, Board on Chemical Sciences and Technology, Commission on Physical Sciences, Mathematics, and Applications, National Research Council (1995). Prudent practices in the laboratory: handling and disposal of chemicals. Washington, D.C.: National Academy Press. ISBN 0-309-05229-7.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ a b Jobelius, Horst H.; Scharff, Hans-Dieter (2005). "Hydrazoic Acid and Azides". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a13_193. ISBN 3527306730.
- ^ Shriver; Atkins. Inorganic Chemistry (5th ed.). New York: W. H. Freeman and Company. p. 382.
- ^ a b c d Rozycki, Michael; Bartha, Richard (1981). "Problems associated with the use of azide as an inhibitor of microbial activity in soil". Applied and Environmental Microbiology. 41 (3): 833–836. doi:10.1128/aem.41.3.833-836.1981. ISSN 0099-2240. PMC 243784. PMID 16345743.
- ^ Lindner, Pinhas; Shomer, Ilan (1984). "Interference of azide in assays of carbohydrates". Food Chemistry. 14 (2): 141–153. doi:10.1016/0308-8146(84)90053-0. ISSN 0308-8146.
- ^ Goel, Ramesh K; Cooper, Adrienne T; Flora, Joseph R.V (2003-09-01). "Sodium azide interference in chemical and biological testing". Journal of Environmental Engineering and Science. 2 (5): 407–411. doi:10.1139/s03-043. ISSN 1496-2551.
- ^ a b Hendrix, Katrien; Bleyen, Nele; Mennecart, Thierry; Bruggeman, Christophe; Valcke, Elie (2019). "Sodium azide used as microbial inhibitor caused unwanted by-products in anaerobic geochemical studies". Applied Geochemistry. 107: 120–130. doi:10.1016/j.apgeochem.2019.05.014. ISSN 0883-2927.
- ^ Aulakh, M. S.; Rennie, D. A. (1985-02-01). "Azide effects upon N2O emission and transformations of N in soils". Canadian Journal of Soil Science. 65 (1): 205–212. doi:10.4141/cjss85-021. ISSN 0008-4271.
- ^ Retelletti Brogi, Simona; Derrien, Morgane; Hur, Jin (2019). "In-depth assessment of the effect of sodium azide on the optical properties of dissolved organic matter". Journal of Fluorescence. 29 (4): 877–885. doi:10.1007/s10895-019-02398-w. ISSN 1053-0509.
- ^ Weber, C. E. (July 1948 – January 1951). "Problems in the use of molten sodium as transfer fluid. OSTI declassified document". Journal of Metallurgy and Ceramics (1–6): 291.
- ^ Huisgen, R. (1961). "Centenary Lecture – 1,3-Dipolar Cycloadditions". Proceedings of the Chemical Society of London: 357. doi:10.1039/PS9610000357.
- ^ H. C. Kolb; M. G. Finn; K. B. Sharpless (2001). "Click Chemistry: Diverse Chemical Function from a Few Good Reactions". Angewandte Chemie International Edition. 40 (11): 2004–2021. doi:10.1002/1521-3773(20010601)40:11<2004::AID-ANIE2004>3.0.CO;2-5. PMID 11433435.
- ^ Kolb, H.C.; Sharpless, B.K. (2003). "The growing impact of click chemistry on drug discovery". Drug Discov Today. 8 (24): 1128–1137. doi:10.1016/S1359-6446(03)02933-7. PMID 14678739.
- ^ Treitler, Daniel S.; Leung, Simon (2 September 2022). "How Dangerous is too Dangerous? A Perspective on Azide Chemistry". The Journal of Organic Chemistry. 87 (17): 11293–11295. doi:10.1021/acs.joc.2c01402. ISSN 0022-3263. PMID 36052475. S2CID 252009657. Retrieved 18 September 2022.
- ^ Mandler, Michael D.; Degnan, Andrew P.; Zhang, Shasha; Aulakh, Darpandeep; Georges, Ketleine; Sandhu, Bhupinder; Sarjeant, Amy; Zhu, Yeheng; Traeger, Sarah C.; Cheng, Peter T.; Ellsworth, Bruce A.; Regueiro-Ren, Alicia (28 January 2022). "Structural and Thermal Characterization of Halogenated Azidopyridines: Under-Reported Synthons for Medicinal Chemistry". Organic Letters. 24 (3): 799–803. doi:10.1021/acs.orglett.1c03201. PMID 34714083. S2CID 240154010.
- ^ Tat, John; Heskett, Karen; Satomi, Shiho; Pilz, Renate B.; Golomb, Beatrice A.; Boss, Gerry R. (2021-08-03). "Sodium azide poisoning: A narrative review". Clinical Toxicology. 59 (8): 683–697. doi:10.1080/15563650.2021.1906888. ISSN 1556-3650. PMC 8349855. PMID 34128439.
- ^ Haas, Jonathan M.; Marsh, William W. (May 1970). "Sodium azide: A potential hazard when used to eliminate interferences in the iodometric determination of sulfur". American Industrial Hygiene Association Journal. 31 (3): 318–321. doi:10.1080/0002889708506248. ISSN 0002-8894.

















