The US FDA’s proposed rule on laboratory-developed tests: Impacts on clinical laboratory testing
Contents
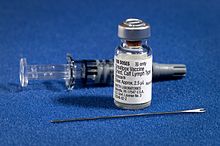 The smallpox vaccine diluent in a syringe alongside a vial of Dryvax dried smallpox vaccine and bifurcated needle | |
| Vaccine description | |
|---|---|
| Target | Smallpox, Mpox |
| Vaccine type | Live virus |
| Clinical data | |
| Trade names | ACAM2000, Imvanex, Jynneos, others |
| AHFS/Drugs.com | Monograph |
| License data | |
| Pregnancy category |
|
| Routes of administration | Subcutaneous,[1][2] percutaneous[3] |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| DrugBank | |
| UNII | |
| KEGG | |
The smallpox vaccine is used to prevent smallpox infection caused by the variola virus.[10] It is the first vaccine to have been developed against a contagious disease. In 1796, British physician Edward Jenner demonstrated that an infection with the relatively mild cowpox virus conferred immunity against the deadly smallpox virus. Cowpox served as a natural vaccine until the modern smallpox vaccine emerged in the 20th century. From 1958 to 1977, the World Health Organization (WHO) conducted a global vaccination campaign that eradicated smallpox,[10] making it the only human disease to be eradicated. Although routine smallpox vaccination is no longer performed on the general public, the vaccine is still being produced for research,[10] and to guard against bioterrorism, biological warfare, and mpox.[11][12]
The term vaccine derives from the Latin word for cow, reflecting the origins of smallpox vaccination. Edward Jenner referred to cowpox as variolae vaccinae (smallpox of the cow). The origins of the smallpox vaccine became murky over time,[13] especially after Louis Pasteur developed laboratory techniques for creating vaccines in the 19th century. Allan Watt Downie demonstrated in 1939 that the modern smallpox vaccine was serologically distinct from cowpox,[14] and vaccinia was subsequently recognized as a separate viral species. Whole-genome sequencing has revealed that vaccinia is most closely related to horsepox, and the cowpox strains found in Great Britain are the least closely related to vaccinia.[15]
Types
As the oldest vaccine, the smallpox vaccine has gone through several generations of medical technology. From 1796 to the 1880s, the vaccine was transmitted from one person to another through arm-to-arm vaccination. Smallpox vaccine was successfully maintained in cattle starting in the 1840s, and calf lymph vaccine became the leading smallpox vaccine in the 1880s. First-generation vaccines grown on the skin of live animals were widely distributed in the 1950s–1970s to eradicate smallpox. Second-generation vaccines were grown in chorioallantoic membrane or cell cultures for greater purity, and they were used in some areas during the smallpox eradication campaign. Third-generation vaccines are based on attenuated strains of vaccinia and saw limited use prior to the eradication of smallpox.[16]
All three generations of vaccine are available in stockpiles. First and second-generation vaccines contain live unattenuated vaccinia virus and can cause serious side effects in a small percentage of recipients, including death in 1–10 people per million vaccinations. Third-generation vaccines are much safer due to the milder side effects of the attenuated vaccinia strains.[16] Second and third-generation vaccines are still being produced, with manufacturing capacity being built up in the 2000s due to fears of bioterrorism and biological warfare.
First-generation

The first-generation vaccines are manufactured by growing live vaccinia virus in the skin of live animals. Most first-generation vaccines are calf lymph vaccines that were grown on the skin of cows, but other animals were also used, including sheep.[16] The development of freeze-dried vaccine in the 1950s made it possible to preserve vaccinia virus for long periods of time without refrigeration, leading to the availability of freeze-dried vaccines such as Dryvax.[18][19]: 115
The vaccine is administered by multiple puncture of the skin (scarification) with a bifurcated needle that holds vaccine solution in the fork.[20] The skin should be cleaned with water rather than alcohol,[20] as the alcohol could inactivate the vaccinia virus.[19]: 292 [21] If alcohol is used, it must be allowed to evaporate completely before the vaccine is administered.[19]: 292 Vaccination results in a skin lesion that fills with pus and eventually crusts over. This manifestation of localized vaccinia infection is known as a vaccine "take" and demonstrates immunity to smallpox. After 2–3 weeks, the scab will fall off and leave behind a vaccine scar.[22]
First generation vaccines consist of live, unattenuated vaccinia virus. One-third of first-time vaccinees develop side effects significant enough to miss school, work, or other activities, or have difficulty sleeping. 15–20% of children receiving the vaccine for the first time develop fevers of over 102 °F (39 °C). The vaccinia lesion can transmit the virus to other people.[22] Rare side effects include postvaccinal encephalitis and myopericarditis.[22][23] Many countries have stockpiled first generation smallpox vaccines. In a 2006 predictive analysis of casualties if there were a mass vaccination of the populations of Germany and the Netherlands, it was estimated that a total of 9.8 people in the Netherlands and 46.2 people in Germany would die from uncontrolled vaccinia infection after being vaccinated with the New York City Board of Health strain. More deaths were predicted for vaccines based other strains: Lister (55.1 Netherlands, 268.5 Germany) and Bern (303.5 Netherlands, 1,381 Germany).[24][25]
Second-generation
The second-generation vaccines consist of live vaccinia virus grown in the chorioallantoic membrane or cell culture. The second-generation vaccines are also administered through scarification with a bifurcated needle, and they carry the same side effects as the first-generation vaccinia strain that was cloned. However, the use of eggs or cell culture allows for vaccine production in a sterile environment, while first-generation vaccine contains skin bacteria from the animal that the vaccine was grown on.[16]
Ernest William Goodpasture, Alice Miles Woodruff, and G. John Buddingh grew vaccinia virus on the chorioallantoic membrane of chicken embryos in 1932.[26] The Texas Department of Health began producing egg-based vaccine in 1939 and started using it in vaccination campaigns in 1948.[19]: 588 Lederle Laboratories began selling its Avianized smallpox vaccine in the United States in 1959.[27] Egg-based vaccine was also used widely in Brazil, New Zealand, and Sweden, and on a smaller scale in many other countries. Concerns about temperature stability and avian sarcoma leukosis virus prevented it from being used more widely during the eradication campaign, although no increase in leukemia was seen in Brazil and Sweden despite the presence of ASLV in the chickens.[19]: 588
Vaccinia was first grown in cell culture in 1931 by Thomas Milton Rivers. The WHO funded work in the 1960s at the Dutch National Institute for Public Health and the Environment (RIVM) on growing the Lister/Elstree strain in rabbit kidney cells and tested it in 45,443 Indonesian children in 1973, with comparable results to the same strain of calf lymph vaccine.[19]: 588–589 Two other cell culture vaccines were developed from the Lister strain in the 2000s: Elstree-BN (Bavarian Nordic) and VV Lister CEP (Chicken Embryo Primary, Sanofi Pasteur).[16][28][29] Lister/Elstree-RIVM was stockpiled in the Netherlands, and Elstree-BN was sold to some European countries for stockpiles.[16] However, Sanofi dropped its own vaccine after it acquired Acambis in 2008.
ACAM2000 is a vaccine developed by Acambis, which was acquired by Sanofi Pasteur in 2008, before selling the smallpox vaccine to Emergent Biosolutions in 2017. Six strains of vaccinia were isolated from 3,000 doses of Dryvax and found to exhibit significant variation in virulence. The strain with the most similar virulence to the overall Dryvax mixture was selected and grown in MRC-5 cells to make the ACAM1000 vaccine. After a successful phase I trial of ACAM1000, the virus was passaged three times in Vero cells to develop ACAM2000, which entered mass production at Baxter. The United States ordered over 200 million doses of ACAM2000 in 1999–2001 for its stockpile, and production is ongoing to replace expired vaccine.[30][31]
ACAM2000 was approved for mpox prevention in the United States in August 2024.[32][33][34]
Third-generation
The third-generation vaccines are based on attenuated vaccinia viruses that are much less virulent and carry lesser side effects. The attenuated viruses may be replicating or non-replicating.[16]
MVA
Modified vaccinia Ankara (MVA, German: Modifiziertes Vakziniavirus Ankara) is a replication-incompetent variant of vaccinia that was developed in West Germany through serial passage. The original Ankara strain of vaccinia was maintained at the vaccine institute in Ankara, Turkey on donkeys and cows. The Ankara strain was taken to West Germany in 1953, where Herrlich and Mayr grew it on chorioallantoic membrane at the University of Munich. After 572 serial passages, the vaccinia virus had lost over 14% of its genome and could no longer replicate in human cells. MVA was used in West Germany in 1977–1980, but the eradication of smallpox ended the vaccination campaign after only 120,000 doses.[35]
MVA stimulates the production of fewer antibodies than replicating vaccines.[36] During the smallpox eradication campaign, MVA was considered to be a pre-vaccine that would be administered before a replicating vaccine to reduce the side effects, or an alternative vaccine that could be safely given to people at high risk from a replicating vaccine.[19]: 585 Japan evaluated MVA and rejected it due to its low immunogenicity, deciding to develop its own attenuated vaccine instead.[37] In the 2000s, MVA was tested in animal models at much higher dosages.[38] When MVA is given to monkeys at 40 times the dosage of Dryvax, it stimulates a more rapid immune response while still causing lesser side effects.[39]
MVA-BN
MVA-BN (also known as: Imvanex in the European Union; Imvamune in Canada; and Jynneos[40][41]) is a vaccine manufactured by Bavarian Nordic by growing MVA in cell culture. Unlike replicating vaccines, MVA-BN is administered by injection via the subcutaneous route and does not result in a vaccine "take."[42] A "take" or "major cutaneous reaction" is a pustular lesion or an area of definite induration or congestion surrounding a central lesion, which can be a scab or an ulcer.[43]
MVA-BN can also be administered intradermally to increase the number of available doses.[44] It is safer for immunocompromised patients and those who are at risk from a vaccinia[definition needed] infection.[citation needed] MVA-BN has been approved in the European Union,[1] Canada,[45][46][47] and the United States.[48][49] Clinical trials have found that MVA-BN is safer and just as immunogenic as ACAM2000.[50][51][52] This vaccine has also been approved for use against mpox.[53][54][55]
LC16m8
LC16m8 is a replicating attenuated strain of vaccinia that is manufactured by Kaketsuken in Japan. Working at the Chiba Serum Institute in Japan, So Hashizume passaged the Lister strain 45 times in primary rabbit kidney cells, interrupting the process after passages 36, 42, and 45 to grow clones on chorioallantoic membrane and select for pock size. The resulting variant was designated LC16m8 (Lister clone 16, medium pocks, clone 8). Unlike the severely-damaged MVA, LC16m8 contains every gene that is present in the ancestral vaccinia. However, a single-nucleotide deletion truncates membrane protein B5R from a residue length of 317 to 92. Although the truncated protein decreases production of extracellular enveloped virus, animal models have shown that antibodies against other membrane proteins are sufficient for immunity. LC16m8 was approved in Japan in 1975 after testing in over 50,000 children. Vaccination with LC16m8 results in a vaccine "take", but safety is similar to MVA.[37]
Safety
Vaccinia is infectious, which improves its effectiveness, but causes serious complications for people with impaired immune systems (for example chemotherapy and AIDS patients) or history of eczema, and is not considered safe for pregnant women.[56] A woman planning on conceiving should not receive smallpox immunization. Vaccines that only contain attenuated vaccinia viruses (an attenuated virus is one in which the pathogenicity has been decreased through serial passage) have been proposed, but some researchers[who?] have questioned the possible effectiveness of such a vaccine. According to the US Centers for Disease Control and Prevention (CDC), "within 3 days of being exposed to the virus, the vaccine might protect you from getting the disease. If you still get the disease, you might get much less sick than an unvaccinated person would. Within 4 to 7 days of being exposed to the virus, the vaccine likely gives you some protection from the disease. If you still get the disease, you might not get as sick as an unvaccinated person would."[57]
In May 2007, the Vaccines and Related Biological Products Advisory Committee (VRBPAC) of the US Food and Drug Administration (FDA) voted unanimously that a new live virus vaccine produced by Acambis, ACAM2000, is both safe and effective for use in persons at high risk of exposure to smallpox virus. However, due to the high rate of serious adverse effects, the vaccine will only be made available to the CDC for the Strategic National Stockpile.[58] ACAM2000 was approved for medical use in the United States in August 2007.[32]
Stockpiles
Since smallpox has been eradicated, the public is not routinely vaccinated against the disease. The World Health Organization maintained a stockpile of 200 million doses in 1980, to guard against reemergence of the disease, but 99% of the stockpile was destroyed in the late 1980s when smallpox failed to return.[16] After the September 11 attacks in 2001, many governments began building up vaccine stockpiles again for fear of bioterrorism. Several companies sold off their stockpiles of vaccines manufactured in the 1970s, and production of smallpox vaccines resumed.[59] Aventis Pasteur discovered a stockpile from the 1950s and donated it to the US government.[60]
Stockpiles of newer vaccines must be repurchased periodically since they carry expiration dates. The United States had received 269 million doses of ACAM2000 and 28 million doses of MVA-BN by 2019,[61][62] but only 100 million doses of ACAM2000 and 65,000 doses of MVA-BN were still available from the stockpile at the start of the 2022 monkeypox outbreak.[63] First-generation vaccines have no specified expiration date and remain viable indefinitely in deep freeze. The U.S. stockpile of WetVax was manufactured in 1956–1957 and maintained since then at −4 °F (−20 °C),[64] and it was still effective when tested in 2004.[65] Replicating vaccines also remain effective even at 1:10 dilution, so a limited number of doses can be stretched to cover a much larger population.[65]
| Country, region, or organization | Year | Doses (millions) | Composition (generation) |
|---|---|---|---|
| 2013 | 2.7 |
| |
(pledged) |
2018 | 27 | Various (1st, 2nd, 3rd)[66][67] |
| 2006 | 55 | 55 million Pourquier (1st)[68] | |
| 2022 | 100 | ||
| 2022 | 5[69] | ||
| 2006 | 56 | LC16m8 (3rd)[37] | |
| 2017 | ? | Lister/Elstree-RIVM (2nd)[19]: 588–589 | |
| 2022 | 35 | Lancy-Vaxina (1st)[70][71] | |
| 2022 | 185 |
History
Variolation
The mortality of the severe form of smallpox – variola major – was very high without vaccination, up to 35% in some outbreaks.[72] A method of inducing immunity known as inoculation, insufflation or "variolation" was practiced before the development of a modern vaccine and likely occurred in Africa and China well before the practice arrived in Europe.[73] It may also have occurred in India, but this is disputed; other investigators contend the ancient Sanskrit medical texts of India do not describe these techniques.[73][74] The first clear reference to smallpox inoculation was made by the Chinese author Wan Quan (1499–1582) in his Douzhen xinfa (痘疹心法) published in 1549.[75] Inoculation for smallpox does not appear to have been widespread in China until the reign era of the Longqing Emperor (r. 1567–1572) during the Ming Dynasty.[76] In China, powdered smallpox scabs were blown up the noses of the healthy. The patients would then develop a mild case of the disease and from then on were immune to it. The technique did have a 0.5–2.0% mortality rate, but that was considerably less than the 20–30% mortality rate of the disease itself. Two reports on the Chinese practice of inoculation were received by the Royal Society in London in 1700; one by Dr. Martin Lister who received a report by an employee of the East India Company stationed in China and another by Clopton Havers.[77] According to Voltaire (1742), the Turks derived their use of inoculation from neighbouring Circassia. Voltaire does not speculate on where the Circassians derived their technique from, though he reports that the Chinese have practiced it "these hundred years".[78]
Variolation was also practiced throughout the latter half of the 17th century by physicians in Turkey, Persia, and Africa. In 1714 and 1716, two reports of the Ottoman Empire Turkish method of inoculation were made to the Royal Society in England, by Emmanuel Timoni, a doctor affiliated with the British Embassy in Constantinople,[79] and Giacomo Pylarini. Source material tells us on Lady Mary Wortley Montagu; "When Lady Mary was in the Ottoman Empire, she discovered the local practice of inoculation against smallpox called variolation."[80] In 1718 she had her son, aged five variolated. He recovered quickly. She returned to London and had her daughter variolated in 1721 by Charles Maitland, during an epidemic of smallpox. This encouraged the British Royal Family to take an interest and a trial of variolation was carried out on prisoners in Newgate Prison. This was successful and in 1722 Caroline of Ansbach, the Princess of Wales, allowed Maitland to vaccinate her children.[81] The success of these variolations assured the British people that the procedure was safe.[79]
...scarred the wrists, legs, and forehead of the patient, placed a fresh and kindly pock in each incision and bound it there for eight or ten days, after this time the patient was credibly informed. The patient would then develop a mild case [of smallpox], recover, and thereafter be immune.[82]
—Dr. Peter Kennedy
Stimulated by a severe epidemic, variolation was first employed in North America in 1721. The procedure had been known in Boston since 1706, when preacher Cotton Mather learned it from Onesimus, a man he held as a slave, who – like many of his peers – had been inoculated in Africa before they were kidnapped.[83] This practice was widely criticized at first.[84] However, a limited trial showed six deaths occurred out of 244 who were variolated (2.5%), while 844 out of 5980 died of natural disease (14%), and the process was widely adopted throughout the colonies.[19]
The inoculation technique was documented as having a mortality rate of only one in a thousand. Two years after Kennedy's description appeared, March 1718, Dr. Charles Maitland successfully inoculated the five-year-old son of the British ambassador to the Turkish court under orders from the ambassador's wife Lady Mary Wortley Montagu, who four years later introduced the practice to England.[85]
An account from letter by Lady Mary Wortley Montagu to Sarah Chiswell, dated 1 April 1717, from the Turkish Embassy describes this treatment:
The small-pox so fatal and so general amongst us is here entirely harmless by the invention of ingrafting (which is the term they give it). There is a set of old women who make it their business to perform the operation. Every autumn in the month of September, when the great heat is abated, people send to one another to know if any of their family has a mind to have the small-pox. They make parties for this purpose, and when they are met (commonly fifteen or sixteen together) the old woman comes with a nutshell full of the matter of the best sort of small-pox and asks what veins you please to have opened. She immediately rips open that you offer to her with a large needle (which gives you no more pain than a common scratch) and puts into the vein as much venom as can lye upon the head of her needle, and after binds up the little wound with a hollow bit of shell, and in this manner opens four or five veins. … The children or young patients play together all the rest of the day and are in perfect health till the eighth. Then the fever begins to seize them and they keep their beds two days, very seldom three. They have very rarely above twenty or thirty in their faces, which never mark, and in eight days time they are as well as before the illness. … There is no example of any one that has died in it, and you may believe I am very well satisfied of the safety of the experiment since I intend to try it on my dear little son. I am patriot enough to take pains to bring this useful invention into fashion in England, and I should not fail to write to some of our doctors very particularly about it if I knew any one of them that I thought had virtue enough to destroy such a considerable branch of their revenue for the good of mankind, but that distemper is too beneficial to them not to expose to all their resentment the hardy wight that should undertake to put an end to it. Perhaps if I live to return I may, however, have courage to war with them.[86]
Early vaccination

In the early empirical days of vaccination, before Louis Pasteur's work on establishing the germ theory and Joseph Lister's on antisepsis and asepsis, there was considerable cross-infection. William Woodville, one of the early vaccinators and director of the London Smallpox Hospital is thought to have contaminated the cowpox matter – the vaccine – with smallpox matter and this essentially produced variolation. Other vaccine material was not reliably derived from cowpox, but from other skin eruptions of cattle.[87]
During the earlier days of empirical experimentation in 1758, American Calvinist Jonathan Edwards died from a smallpox inoculation. Some of the earliest statistical and epidemiological studies were performed by James Jurin in 1727 and Daniel Bernoulli in 1766.[88] In 1768, Dr John Fewster reported that variolation induced no reaction in persons who had had cowpox.[89][90]

Edward Jenner was born in Berkeley, England. As a young child, Jenner was variolated with the other schoolboys through parish funds, but nearly died due to the seriousness of his infection. Fed purgative medicine and going through the bloodletting process, Jenner was put in one of the variolation stables until he recovered.[91] At the age of 13, he was apprenticed to apothecary Daniel Ludlow and later surgeon George Hardwick in nearby Sodbury. He observed that people who caught cowpox while working with cattle were known not to catch smallpox. Jenner assumed a causal connection but the idea was not taken up at that time. From 1770 to 1772 Jenner received advanced training in London at St. George's Hospital and as the private pupil of John Hunter, then returned to set up practice in Berkeley.[92]
Perhaps there was already an informal public understanding of some connection between disease resistance and working with cattle. The "beautiful milkmaid" seems to have been a frequent image in the art and literature of this period. But it is known for certain that in the years following 1770, at least six people in England and Germany (Sevel, Jensen, Jesty 1774, Rendall, Plett 1791) tested successfully the possibility of using the cowpox vaccine as an immunization for smallpox in humans.[93]

Jenner sent a paper reporting his observations to the Royal Society in April 1797. It was not submitted formally and there is no mention of it in the Society's records. Jenner had sent the paper informally to Sir Joseph Banks, the Society's president, who asked Everard Home for his views. Reviews of his rejected report, published for the first time in 1999, were skeptical and called for further vaccinations.[94] Additional vaccinations were performed and in 1798 Jenner published his work entitled An Inquiry into the Causes and Effects of the Variolae Vaccinae, a disease discovered in some of the western counties of England, particularly Gloucestershire and Known by the Name of Cow Pox.[73][95][96] It was an analysis of 23 cases including several individuals who had resisted natural exposure after previous cowpox. It is not known how many Jenner vaccinated or challenged by inoculation with smallpox virus; e.g. Case 21 included 'several children and adults'. Crucially all of at least four whom Jenner deliberately inoculated with smallpox virus resisted it. These included the first and last patients in a series of arm-to-arm transfers. He concluded that cowpox inoculation was a safe alternative to smallpox inoculation, but rashly claimed that the protective effect was lifelong. This last proved to be incorrect.[97] Jenner also tried to distinguish between 'True' cowpox which produced the desired result and 'Spurious' cowpox which was ineffective and/or produced severe reaction. Modern research suggests Jenner was trying to distinguish between effects caused by what would now[when?] be recognised as non-infectious vaccine, a different virus (e.g. paravaccinia/milker's nodes), or contaminating bacterial pathogens. This caused confusion at the time, but would become important criteria in vaccine development.[98] A further source of confusion was Jenner's belief that fully effective vaccine obtained from cows originated in an equine disease, which he mistakenly referred to as grease. This was criticised at the time but vaccines derived from horsepox were soon introduced and later contributed to the complicated problem of the origin of vaccinia virus, the virus in present-day vaccine.[99]: 165–78
The introduction of the vaccine to the New World took place in Trinity, Newfoundland, in 1798 by Dr. John Clinch, boyhood friend and medical colleague of Jenner.[100][101] The first smallpox vaccine in the United States was administered in 1799. The physician Valentine Seaman gave his children a smallpox vaccination using a serum acquired from Jenner.[102][103] By 1800, Jenner's work had been published in all the major European languages and had reached Benjamin Waterhouse in the United States – an indication of rapid spread and deep interest.[104]: 262–67 Despite some concern about the safety of vaccination the mortality using carefully selected vaccine was close to zero, and it was soon in use all over Europe and the United States.[105][106]
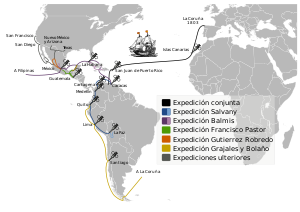
In 1804 the Balmis Expedition, an official Spanish mission commanded by Francisco Javier de Balmis, sailed to spread the vaccine throughout the Spanish Empire, first to the Canary Islands and on to Spanish Central America. While his deputy, José Salvany, took vaccine to the west and east coasts of Spanish South America, Balmis sailed to Manila in the Philippines and on to Canton and Macao on the Chinese coast. He returned to Spain in 1806.[107] The vaccine was not carried in the form of flasks, but in the form of 22 orphaned boys, who were 'carriers' of the live cowpox virus. After arrival, "other Spanish governors and doctors used enslaved girls to move the virus between islands, using lymph fluid harvested from them to inoculate their local populations".[108]
Napoleon was an early proponent of smallpox vaccination and ordered that army recruits be given the vaccine. Additionally a vaccination program was created for the French Army and his Imperial Guard. In 1811 he had his son, Napoleon II, vaccinated after his birth. By 1815 about half of French children were vaccinated and by the end of the Napoleonic Empire smallpox deaths accounted for 1.8% of deaths, as opposed to the 4.8% of deaths it accounted for at the time of the French Revolution.[109]
The first state to introduce compulsory vaccinations was the Principality of Lucca and Piombino on 25 September 1806.[110] On 26 August 1807, Bavaria introduced a similar measure. Baden followed in 1809, Prussia in 1815, Württemberg in 1818, Sweden in 1816, England in 1867 and the German Empire in 1874 through the Reichs Vaccination Act.[111][112] In Lutheran Sweden, the Protestant clergy played a pioneering role in voluntary smallpox vaccination as early as 1800.[113] The first vaccination was carried out in Liechtenstein in 1801, and from 1812 it was mandatory to vaccinate.[114]
The question of who first tried cowpox inoculation/vaccination cannot be answered with certainty. Most, but still limited, information is available for Benjamin Jesty, Peter Plett and John Fewster. In 1774 Jesty, a farmer of Yetminster in Dorset, observing that the two milkmaids living with his family were immune to smallpox, inoculated his family with cowpox to protect them from smallpox. He attracted a certain amount of local criticism and ridicule at the time then interest waned. Attention was later drawn to Jesty, and he was brought to London in 1802 by critics jealous of Jenner's prominence at a time when he was applying to Parliament for financial reward.[115] During 1790–92 Peter Plett, a teacher from Holstein, reported limited results of cowpox inoculation to the Medical Faculty of the University of Kiel. However, the Faculty favoured variolation and took no action.[116] John Fewster, a surgeon friend of Jenner's from nearby Thornbury, discussed the possibility of cowpox inoculation at meetings as early as 1765. He may have done some cowpox inoculations in 1796 at about the same time that Jenner vaccinated Phipps. However, Fewster, who had a flourishing variolation practice, may have considered this option but used smallpox instead. He thought vaccination offered no advantage over variolation, but maintained friendly contact with Jenner and certainly made no claim of priority for vaccination when critics attacked Jenner's reputation.[117] It seems clear that the idea of using cowpox instead of smallpox for inoculation was considered, and actually tried in the late 18th century, and not just by the medical profession. Therefore, Jenner was not the first to try cowpox inoculation. However, he was the first to publish his evidence and distribute vaccine freely, provide information on selection of suitable material, and maintain it by arm-to-arm transfer. The authors of the official World Health Organization (WHO) account Smallpox and its Eradication assessing Jenner's role wrote:[19]: 264
Publication of the Inquiry and the subsequent energetic promulgation by Jenner of the idea of vaccination with a virus other than variola virus constituted a watershed in the control of smallpox for which he, more than anyone else deserves the credit.
As vaccination spread, some European countries made it compulsory. Concern about its safety led to opposition and then repeal of legislation in some instances.[117]: 236–40 [118] Compulsory infant vaccination was introduced in England by the Vaccination Act 1853 (16 & 17 Vict. c. 100). By 1871, parents could be fined for non-compliance, and then imprisoned for non-payment.[118]: 202–13 This intensified opposition, and the Vaccination Act 1898 (61 & 62 Vict. c. 49) introduced a conscience clause.[119] This allowed exemption on production of a certificate of conscientious objection signed by two magistrates. Such certificates were not always easily obtained and a further act in 1907 allowed exemption by a statutory declaration which could not be refused. Although theoretically still compulsory, the Vaccination Act 1907 (7 Edw. 7. c. 31) effectively marked the end of compulsory infant vaccination in England.[118]: 233–38

In the United States vaccination was regulated by individual states, the first to impose compulsory vaccination being Massachusetts in 1809. There then followed sequences of compulsion, opposition and repeal in various states. By 1930 Arizona, Utah, North Dakota and Minnesota prohibited compulsory vaccination, 35 states allowed regulation by local authorities, or had no legislation affecting vaccination, whilst in ten states, including Washington, D.C. and Massachusetts, infant vaccination was compulsory.[104]: 292–93 Compulsory infant vaccination was regulated by only allowing access to school for those who had been vaccinated.[120] Those seeking to enforce compulsory vaccination argued that the public good overrode personal freedom, a view supported by the U.S. Supreme Court in Jacobson v. Massachusetts in 1905, a landmark ruling which set a precedent for cases dealing with personal freedom and the public good.[121]
Louis T. Wright,[122] an African-American Harvard Medical School graduate (1915), introduced, while serving in the Army during World War I, intradermal, smallpox vaccination for the soldiers.[123]
Developments in production
Until the end of the 19th century, vaccination was performed either directly with vaccine produced on the skin of calves or, particularly in England, with vaccine obtained from the calf but then maintained by arm-to-arm transfer;[124] initially in both cases vaccine could be dried on ivory points for short-term storage or transport but increasing use was made of glass capillary tubes for this purpose towards the end of the century.[125] During this period there were no adequate methods for assessing the safety of the vaccine and there were instances of contaminated vaccine transmitting infections such as erysipelas, tetanus, septicaemia and tuberculosis.[98] In the case of arm-to-arm transfer there was also the risk of transmitting syphilis. Although this did occur occasionally, estimated as 750 cases in 100 million vaccinations,[105]: 122 some critics of vaccination e.g. Charles Creighton believed that uncontaminated vaccine itself was a cause of syphilis.[126] Smallpox vaccine was the only vaccine available during this period, and so the determined opposition to it initiated a number of vaccine controversies that spread to other vaccines and into the 21st century.[citation needed]
Sydney Arthur Monckton Copeman, an English Government bacteriologist interested in smallpox vaccine investigated the effects on the bacteria in it of various treatments, including glycerine. Glycerine was sometimes used simply as a diluent by some continental vaccine producers. However, Copeman found that vaccine suspended in 50% chemically pure glycerine and stored under controlled conditions contained very few "extraneous" bacteria and produced satisfactory vaccinations.[127] He later reported that glycerine killed the causative organisms of erysipelas and tuberculosis when they were added to the vaccine in "considerable quantity", and that his method was widely used on the continent.[124] In 1896, Copeman was asked to supply "extra good calf vaccine" to vaccinate the future Edward VIII.[128]
Vaccine produced by Copeman's method was the only type issued free to public vaccinators by the British Government Vaccine Establishment from 1899. At the same time the Vaccination Act 1898 (61 & 62 Vict. c. 49) banned arm-to-arm vaccination, thus preventing transmission of syphilis by this vaccine. However, private practitioners had to purchase vaccine from commercial producers.[129] Although proper use of glycerine reduced bacterial contamination considerably the crude starting material, scraped from the skin of infected calves, was always heavily contaminated and no vaccine was totally free from bacteria. A survey of vaccines in 1900 found wide variations in bacterial contamination. Vaccine issued by the Government Vaccine Establishment contained 5,000 bacteria per gram, while commercial vaccines contained up to 100,000 per gram.[130] The level of bacterial contamination remained unregulated until the Therapeutic Substances Act 1925 (15 & 16 Geo. 5. c. 60) set an upper limit of 5,000 per gram, and rejected any batch of vaccine found to contain the causative organisms of erysipelas or wound infections.[98] Unfortunately glycerolated vaccine lost its potency quickly at ambient temperatures which restricted its use in tropical climates. However, it remained in use into the 1970s when a satisfactory cold chain was available. Animals continued to be widely used by vaccine producers during the smallpox eradication campaign. A WHO survey of 59 producers, some of whom used more than one source of vaccine, found that 39 used calves, 12 used sheep and 6 used water buffalo, whilst only 3 made vaccine in cell culture and 3 in embryonated hens' eggs.[19]: 543–45 English vaccine was occasionally made in sheep during World War I but from 1946 only sheep were used.[125]
In the late 1940s and early 1950s, Leslie Collier, an English microbiologist working at the Lister Institute of Preventive Medicine, developed a method for producing a heat-stable freeze-dried vaccine in powdered form.[131][132] Collier added 0.5% phenol to the vaccine to reduce the number of bacterial contaminants but the key stage was to add 5% peptone to the liquid vaccine before it was dispensed into ampoules. This protected the virus during the freeze drying process. After drying, the ampoules were sealed under nitrogen. Like other vaccines, once reconstituted it became ineffective after 1–2 days at ambient temperatures. However, the dried vaccine was 100% effective when reconstituted after 6 months storage at 37 °C (99 °F) allowing it to be transported to, and stored in, remote tropical areas. Collier's method was increasingly used and, with minor modifications, became the standard for vaccine production adopted by the WHO Smallpox Eradication Unit when it initiated its global smallpox eradication campaign in 1967, at which time 23 of 59 manufacturers were using the Lister strain.[19]: 545, 550
In a letter about landmarks in the history of smallpox vaccine, written to and quoted from by Derrick Baxby, Donald Henderson, chief of the Smallpox Eradication Unit from 1967 to 1977 wrote; "Copeman and Collier made an enormous contribution for which neither, in my opinion ever received due credit".[133]
Smallpox vaccine was inoculated by scratches into the superficial layers of the skin, with a wide variety of instruments used to achieve this. They ranged from simple needles to multi-pointed and multi-bladed spring-operated instruments specifically designed for the purpose.[134]
A major contribution to smallpox vaccination was made in the 1960s by Benjamin Rubin, an American microbiologist working for Wyeth Laboratories. Based on initial tests with textile needles with the eyes cut off transversely half-way he developed the bifurcated needle. This was a sharpened two-prong fork designed to hold one dose of reconstituted freeze-dried vaccine by capillarity.[135] Easy to use with minimum training, cheap to produce ($5 per 1000), using one quarter as much vaccine as other methods, and repeatedly re-usable after flame sterilization, it was used globally in the WHO Smallpox Eradication Campaign from 1968.[19]: 472–73, 568–72 Rubin estimated that it was used to do 200 million vaccinations per year during the last years of the campaign.[135] Those closely involved in the campaign were awarded the "Order of the Bifurcated Needle". This, a personal initiative by Donald Henderson, was a lapel badge, designed and made by his daughter, formed from the needle shaped to form an "O". This represented "Target Zero", the objective of the campaign.[136]
Eradication of smallpox

Smallpox was eradicated by a massive international search for outbreaks, backed up with a vaccination program, starting in 1967. It was organised and co-ordinated by a World Health Organization (WHO) unit, set up and headed by Donald Henderson. The last case in the Americas occurred in 1971 (Brazil), south-east Asia (Indonesia) in 1972, and on the Indian subcontinent in 1975 (Bangladesh). After two years of intensive searches, what proved to be the last endemic case anywhere in the world occurred in Somalia, in October 1977.[19]: 526–37 A Global Commission for the Certification of Smallpox Eradication chaired by Frank Fenner examined the evidence from, and visited where necessary, all countries where smallpox had been endemic. In December 1979 they concluded that smallpox had been eradicated; a conclusion endorsed by the WHO General Assembly in May 1980.[19]: 1261–62 However, even as the disease was being eradicated there still remained stocks of smallpox virus in many laboratories. Accelerated by two cases of smallpox in 1978, one fatal (Janet Parker), caused by an accidental and unexplained containment breach at a laboratory at the University of Birmingham Medical School, the WHO ensured that known stocks of smallpox virus were either destroyed or moved to safer laboratories. By 1979, only four laboratories were known to have smallpox virus. All English stocks held at St Mary's Hospital, London were transferred to more secure facilities at Porton Down and then to the US at the Centers for Disease Control and Prevention (CDC) in Atlanta, Georgia in 1982, and all South African stocks were destroyed in 1983. By 1984, the only known stocks were kept at the CDC in the U.S. and the State Research Center of Virology and Biotechnology (VECTOR) in Koltsovo, Russia.[19]: 1273–76 These states report that their repositories are for possible anti-bioweaponry research and insurance if some obscure reservoir of natural smallpox is discovered in the future.[citation needed][137]
Anti-terrorism preparation
Among more than 270,000 US military service members vaccinated with smallpox vaccine between December 2002, and March 2003, eighteen cases of probable myopericarditis were reported (all in first-time vaccinees who received the NYCBOH strain of vaccinia virus), an incidence of 7.8 per 100,000 during the 30 days they were observed. All cases were in young, otherwise healthy adult white men and all survived.[138]
In 2002, the United States government started a program to vaccinate 500,000 volunteer health care professionals throughout the country. Recipients were healthcare workers who would be first-line responders in the event of a bioterrorist attack. Many healthcare workers refused or did not pursue vaccination, worried about vaccine side effects, compensation and liability. Most did not see an immediate need for the vaccine. Some healthcare systems refused to participate, worried about becoming a destination for smallpox patients in the event of an epidemic.[139] Fewer than 40,000 actually received the vaccine.[140]
On 21 April 2022, Public Services and Procurement Canada published a notice of tender seeking to stockpile 500,000 doses of smallpox vaccine in order to protect against a potential accidental or intentional release of the eradicated virus.[141] On 6 May, the contract was awarded to Bavarian Nordic for their Imvamune vaccine.[142] These were deployed by the Public Health Agency of Canada for targeted vaccination in response to the 2022 mpox outbreak.[143]
Origin
The origin of the modern smallpox vaccine has long been unclear,[144] but horsepox was identified in the 2010s as the most likely ancestor.[145]: 9 Edward Jenner had obtained his vaccine from a cow, so he named the virus vaccinia, after the Latin word for cow. Jenner believed that both cowpox and smallpox were viruses that originated in the horse and passed to the cow,[146]: 52–53 and some doctors followed his reasoning by inoculating their patients directly with horsepox.[147] The situation was further muddied when Louis Pasteur developed techniques for creating vaccines in the laboratory in the late 19th century. As medical researchers subjected viruses to serial passage, inadequate recordkeeping resulted in the creation of laboratory strains with unclear origins.[99]: 4 By the late 19th century, it was unknown whether the vaccine originated from cowpox, horsepox, or an attenuated strain of smallpox.[148]
In 1939, Allan Watt Downie showed that the vaccinia virus was serologically distinct from the "spontaneous" cowpox virus.[14] This work established vaccinia and cowpox as two separate viral species. The term vaccinia now refers only to the smallpox vaccine,[149] while cowpox no longer has a Latin name.[150] The development of whole genome sequencing in the 1990s made it possible to compare orthopoxvirus genomes and identify their relationships with each other. The horsepox virus was sequenced in 2006 and found to be most closely related to vaccinia.[151] In a phylogenetic tree of the orthopoxviruses, horsepox forms a clade with vaccinia strains, and cowpox strains form a different clade.[15]
Horsepox is extinct in the wild, and the only known sample was collected in 1976.[152] Because the sample was collected at the end of the smallpox eradication campaign, scientists considered the possibility that horsepox is a strain of vaccinia that had escaped into the wild.[153] However, as more smallpox vaccines were sequenced, older vaccines were found to be more similar to horsepox than modern vaccinia strains. A smallpox vaccine manufactured by Mulford in 1902 is 99.7% similar to horsepox, closer than any previously known strain of vaccinia.[154] Modern Brazilian vaccines with a documented introduction date of 1887, made from material collected in an 1866 outbreak of "cowpox" in France, are more similar to horsepox than other strains of vaccinia.[155] Five smallpox vaccines manufactured in the United States in 1859–1873 are most similar to each other and horsepox,[153] as well as the 1902 Mulford vaccine.[156] One of the 1859–1873 vaccines was identified as a novel strain of horsepox, containing a complete gene from the 1976 horsepox sample that has deletions in vaccinia.[156]
Terminology
The word "vaccine" is derived from Variolae vaccinae (i.e. smallpox of the cow), the term devised by Jenner to denote cowpox and used in the long title of his An enquiry into the causes and effects of Variolae vaccinae, known by the name of cow pox.[97] Vaccination, the term which soon replaced cowpox inoculation and vaccine inoculation, was first used in print by Jenner's friend, Richard Dunning in 1800.[92] Initially, the terms vaccine/vaccination referred only to smallpox, but in 1881 Louis Pasteur proposed at the 7th International Congress of Medicine[157] that to honour Jenner the terms be widened to cover the new protective inoculations being introduced.[158] According to some sources the term was first introduced by Jenner's friend Richard Dunning in 1800.[159]
References
- ^ a b c "Imvanex EPAR". European Medicines Agency (EMA). 16 August 2013. Archived from the original on 27 April 2022. Retrieved 2 October 2014.
- ^ a b "Jynneos- vaccinia virus modified strain ankara-bavarian nordic non-replicating antigen injection, suspension". DailyMed. 14 February 2022. Archived from the original on 27 May 2022. Retrieved 26 May 2022.
- ^ a b "ACAM2000 (smallpox- vaccinia vaccine, live injection, powder, lyophilized, for solution)". DailyMed. 21 March 2018. Archived from the original on 24 May 2022. Retrieved 26 May 2022.
- ^ "ACAM2000 smallpox vaccine (live vaccinia virus) vial with diluent". Therapeutic Goods Administration (TGA). Archived from the original on 18 April 2015. Retrieved 8 July 2022.
- ^ "Imvamune Product information". Health Canada. 25 April 2012. Archived from the original on 22 June 2021. Retrieved 8 July 2022.
- ^ "Smallpox vaccine Product information". Health Canada. 25 April 2012. Archived from the original on 8 July 2022. Retrieved 8 July 2022.
- ^ "Smallpox vaccine Product information". Health Canada. 25 April 2012. Archived from the original on 8 July 2022. Retrieved 8 July 2022.
- ^ "Regulatory Decision Summary for ACAM2000". Drug and Health Products Portal. 5 December 2023. Archived from the original on 2 April 2024. Retrieved 2 April 2024.
- ^ "Jynneos Smallpox and Monkeypox Vaccine" (PDF). Archived (PDF) from the original on 3 September 2023. Retrieved 2 September 2023.
- ^ a b c World Health Organization (August 2024). "Smallpox and mpox (orthopoxviruses) vaccine position paper". Weekly Epidemiological Record. 99 (34): 429–456. hdl:10665/378526.
- ^ Metzger W, Mordmueller BG (July 2007). Metzger W (ed.). "Vaccines for preventing smallpox". The Cochrane Database of Systematic Reviews. 2007 (3): CD004913. doi:10.1002/14651858.CD004913.pub2. PMC 6532594. PMID 17636779.
- ^ Anderson MG, Frenkel LD, Homann S, and Guffey J. (2003), "A case of severe monkeypox virus disease in an American child: emerging infections and changing professional values"; Pediatr Infect Dis J;22(12): 1093–96; discussion 1096–98.
- ^ Baxby D (1981). Jenner's Smallpox Vaccine: The Riddle of Vaccinia Virus and Its Origin. Heinemann Educational Books. ISBN 978-0-435-54057-9.
- ^ a b Downie AW (April 1939). "The Immunological Relationship of the Virus of Spontaneous Cowpox to Vaccinia Virus". British Journal of Experimental Pathology. 20 (2): 158–76. ISSN 0007-1021. PMC 2065307.
- ^ a b Carroll DS, Emerson GL, Li Y, Sammons S, Olson V, Frace M, et al. (August 2011). "Chasing Jenner's vaccine: revisiting cowpox virus classification". PLOS ONE. 6 (8): e23086. Bibcode:2011PLoSO...623086C. doi:10.1371/journal.pone.0023086. PMC 3152555. PMID 21858000.
- ^ a b c d e f g h Operational framework for the deployment of the World Health Organization Smallpox Vaccine Emergency Stockpile in response to a smallpox event. World Health Organization (WHO). 2017. ISBN 978-92-4-151341-8.
- ^ "1613.002 | Collections Online". collections.thackraymuseum.co.uk. Retrieved 29 May 2024.
- ^ Belongia EA, Naleway AL (April 2003). "Smallpox vaccine: the good, the bad, and the ugly". Clinical Medicine & Research. 1 (2): 87–92. doi:10.3121/cmr.1.2.87. PMC 1069029. PMID 15931293.
- ^ a b c d e f g h i j k l m n o p Fenner F, Henderson DA, Arita I, Jezek Z, Ladnyi ID (1988). Smallpox and Its Eradication (PDF). History of International Public Health. Geneva: World Health Organization (WHO). hdl:10665/39485. ISBN 978-92-4-156110-5. Archived (PDF) from the original on 25 May 2021. Retrieved 5 November 2013.
- ^ a b "Instructions for smallpox vaccination with bifurcated needle". World Health Organization (WHO). 1968. Archived from the original on 16 March 2022. Retrieved 3 July 2022.
- ^ "Six bifurcated needles for smallpox vaccination | Science Museum Group Collection". Science Museum (London). Archived from the original on 20 May 2022. Retrieved 3 July 2022.
The skin was not disinfected before use as this killed the vaccine.
- ^ a b c Cono J, Casey CG, Bell DM (February 2003). "Smallpox vaccination and adverse reactions. Guidance for clinicians" (PDF). MMWR. Recommendations and Reports. 52 (RR-4): 1–28. PMID 12617510. Archived (PDF) from the original on 22 March 2022. Retrieved 11 August 2022.
- ^ Cassimatis DC, Atwood JE, Engler RM, Linz PE, Grabenstein JD, Vernalis MN (May 2004). "Smallpox vaccination and myopericarditis: a clinical review". Journal of the American College of Cardiology. 43 (9): 1503–1510. doi:10.1016/j.jacc.2003.11.053. PMID 15120802.
- ^ Kretzschmar M, Wallinga J, Teunis P, Xing S, Mikolajczyk R (August 2006). "Frequency of adverse events after vaccination with different vaccinia strains". PLOS Medicine. 3 (8): e272. doi:10.1371/journal.pmed.0030272. PMC 1551910. PMID 16933957.
- ^ Kretzschmar M, Wallinga J, Teunis P, Xing S, Mikolajczyk R (3 October 2006). "Correction: Frequency of Adverse Events after Vaccination with Different Vaccinia Strains". PLOS Medicine. 3 (10): e429. doi:10.1371/journal.pmed.0030429. PMC 1626554.
- ^ Goodpasture EW, Woodruff AM, Buddingh GJ (May 1932). "Vaccinal Infection of the Chorio-Allantoic Membrane of the Chick Embryo". The American Journal of Pathology. 8 (3): 271–282.7. PMC 2062681. PMID 19970016.
- ^ Council on Drugs (American Medical Association) (1964). New and Nonofficial Drugs. Lippincott. p. 739.
- ^ Wiser I, Balicer RD, Cohen D (January 2007). "An update on smallpox vaccine candidates and their role in bioterrorism related vaccination strategies". Vaccine. 25 (6): 976–984. doi:10.1016/j.vaccine.2006.09.046. PMID 17074424.
- ^ Ferrier-Rembert A, Drillien R, Meignier B, Garin D, Crance JM (November 2007). "Safety, immunogenicity and protective efficacy in mice of a new cell-cultured Lister smallpox vaccine candidate". Vaccine. 25 (49): 8290–8297. doi:10.1016/j.vaccine.2007.09.050. PMID 17964011.
- ^ Monath TP, Caldwell JR, Mundt W, Fusco J, Johnson CS, Buller M, et al. (October 2004). "ACAM2000 clonal Vero cell culture vaccinia virus (New York City Board of Health strain)--a second-generation smallpox vaccine for biological defense". International Journal of Infectious Diseases. 8 (Suppl 2): S31–S44. doi:10.1016/j.ijid.2004.09.002. PMC 7110559. PMID 15491873.
- ^ Nalca A, Zumbrun EE (May 2010). "ACAM2000: the new smallpox vaccine for United States Strategic National Stockpile". Drug Design, Development and Therapy. 4: 71–79. doi:10.2147/dddt.s3687. PMC 2880337. PMID 20531961.
- ^ a b "ACAM2000". U.S. Food and Drug Administration (FDA). 29 August 2024. Archived from the original on 17 October 2019. Retrieved 1 September 2024.
- ^ "FDA Roundup: August 30, 2024". U.S. Food and Drug Administration (FDA) (Press release). 30 August 2024. Archived from the original on 1 September 2024. Retrieved 1 September 2024.
- ^ "Emergent Biosolutions' ACAM2000, (Smallpox and Mpox (Vaccinia) Vaccine, Live) Receives U.S. FDA Approval for Mpox Indication; Public Health Mpox Outbreak Continues Across Africa & Other Regions" (Press release). Emergent Biosolutions. 29 August 2024. Archived from the original on 1 September 2024. Retrieved 1 September 2024 – via GlobeNewswire.
- ^ Volz A, Sutter G (2017). "Modified Vaccinia Virus Ankara: History, Value in Basic Research, and Current Perspectives for Vaccine Development". Advances in Virus Research. 97: 187–243. doi:10.1016/bs.aivir.2016.07.001. ISBN 9780128118016. PMC 7112317. PMID 28057259.
- ^ Mayr A, Hochstein-Mintzel V, Stickl H (March 1975). "Abstammung, Eigenschaften und Verwendung des attenuierten Vaccinia-Stammes MVA". Infection. 3 (1): 6–14. doi:10.1007/BF01641272. S2CID 46979748.
- ^ a b c Kenner J, Cameron F, Empig C, Jobes DV, Gurwith M (November 2006). "LC16m8: an attenuated smallpox vaccine". Vaccine. 24 (47–48): 7009–7022. doi:10.1016/j.vaccine.2006.03.087. PMC 7115618. PMID 17052815.
- ^ Cohen J (1 July 2022). "There's a shortage of monkeypox vaccine. Could one dose instead of two suffice?". AAAS. Archived from the original on 8 July 2022. Retrieved 3 July 2022.
Because MVA does not make copies of itself, the team gave it at a higher dose—similar to what's used in the Bavarian Nordic shot today—than the Dryvax vaccine.
- ^ Earl PL, Americo JL, Wyatt LS, Espenshade O, Bassler J, Gong K, et al. (August 2008). "Rapid protection in a monkeypox model by a single injection of a replication-deficient vaccinia virus". Proceedings of the National Academy of Sciences of the United States of America. 105 (31): 10889–10894. Bibcode:2008PNAS..10510889E. doi:10.1073/pnas.0804985105. PMC 2495015. PMID 18678911.
- ^ "Smallpox Vaccine Supply & Strength". National Institute of Allergy and Infectious Diseases (NIAID). 26 September 2019. Archived from the original on 17 October 2019. Retrieved 16 October 2019.
- ^ Greenberg RN, Hay CM, Stapleton JT, Marbury TC, Wagner E, Kreitmeir E, et al. (2016). "A Randomized, Double-Blind, Placebo-Controlled Phase II Trial Investigating the Safety and Immunogenicity of Modified Vaccinia Ankara Smallpox Vaccine (MVA-BN) in 56-80-Year-Old Subjects". PLOS ONE. 11 (6): e0157335. Bibcode:2016PLoSO..1157335G. doi:10.1371/journal.pone.0157335. PMC 4915701. PMID 27327616.
- ^ "Summary Basis for Regulatory Action Template". U.S. Food & Drug Administration. Archived from the original on 8 October 2021. Retrieved 8 October 2021.
- ^ "Vaccine "Take" Evaluation". U.S. Centers for Disease Control and Prevention (CDC). Archived from the original on 26 January 2022. Retrieved 9 January 2022.
- ^ "Monkeypox in the U.S." U.S. Centers for Disease Control and Prevention (CDC). 13 October 2022. Archived from the original on 19 October 2022. Retrieved 19 October 2022.
- ^ "Smallpox and monkeypox vaccine: Canadian Immunization Guide". Public Health Agency of Canada. 16 June 2022. Archived from the original on 20 July 2020. Retrieved 8 July 2022.
- ^ "Register of Innovative Drugs" (PDF). Health Canada. June 2020. Archived (PDF) from the original on 26 June 2020. Retrieved 24 June 2020.
- ^ "Products for Human Use. Submission #144762". Register of Innovative Drugs. Health Canada. Archived from the original on 17 June 2014. Retrieved 30 October 2014.
- ^ "Jynneos". U.S. Food and Drug Administration (FDA). 24 September 2019. STN 125678. Archived from the original on 17 October 2019. Retrieved 16 October 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "FDA approves first live, non-replicating vaccine to prevent smallpox and monkeypox". U.S. Food and Drug Administration (FDA) (Press release). 24 September 2019. Archived from the original on 17 October 2019. Retrieved 17 October 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "Infectious Diseases: Clinical Trials". Bavarian Nordic. Archived from the original on 26 April 2016. Retrieved 30 October 2014.
- ^ "Phase II Trial to Assess Safety and Immunogenicity of Imvamune". ClinicalTrials.gov. U.S. National Institutes of Health. Archived from the original on 13 March 2022. Retrieved 30 October 2014.
- ^ Pittman PR, Hahn M, Lee HS, Koca C, Samy N, Schmidt D, et al. (November 2019). "Phase 3 Efficacy Trial of Modified Vaccinia Ankara as a Vaccine against Smallpox". The New England Journal of Medicine. 381 (20): 1897–1908. doi:10.1056/NEJMoa1817307. PMID 31722150.
- ^ "Jynneos Vaccine Effectiveness". U.S. Centers for Disease Control and Prevention (CDC). 19 May 2023. Archived from the original on 24 May 2023. Retrieved 24 May 2023.
- ^ "Considerations on posology for the use of the vaccine Jynneos/ Imvanex (MVA-BN) against monkeypox" (PDF). European Medicines Agency. 19 August 2022. Archived (PDF) from the original on 28 May 2023. Retrieved 28 May 2023.
- ^ "Protecting you from mpox (monkeypox): information on the smallpox vaccination". GOV.UK. Archived from the original on 28 May 2023. Retrieved 28 May 2023.
- ^ Rotz LD, Dotson DA, Damon IK, Becher JA (June 2001). "Vaccinia (smallpox) vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2001" (PDF). MMWR. Recommendations and Reports. 50 (RR-10): 1–25, quiz CE1–7. PMID 15580803. Archived (PDF) from the original on 13 October 2021. Retrieved 13 June 2022.
- ^ "Prevention and Treatment". U.S. Centers for Disease Control and Prevention (CDC). 18 October 2021. Archived from the original on 15 December 2017. Retrieved 11 August 2022.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "Vaccines and Related Biological Products Advisory Committee Meeting". U.S. Food and Drug Administration (FDA). 17 May 2007. Archived from the original on 20 October 2017. Retrieved 2 May 2013.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ a b c d Kneip A (26 January 2003). "Pocken-Fieber". Der Spiegel (in German). Archived from the original on 4 July 2022. Retrieved 4 July 2022.
- ^ Lueck S (1 April 2002). "Aventis to Donate Smallpox Vaccine To the U.S. Government's Stockpile". The Wall Street Journal. Archived from the original on 12 January 2018. Retrieved 4 July 2022.
- ^ Emergent BioSolutions (3 September 2019). "Emergent BioSolutions Awarded 10-Year HHS Contract to Deliver ACAM2000, (Smallpox (Vaccinia) Vaccine, Live) Into the Strategic" (Press release). Archived from the original on 7 November 2023. Retrieved 4 July 2022.
- ^ Bavarian Nordic. "Bavarian Nordic Announces U.S. FDA Approval of Jynneos (Smallpox and Monkeypox Vaccine, Live, Non-Replicating) for Prevention of Smallpox and Monkeypox Disease in Adults" (Press release). Archived from the original on 28 June 2022. Retrieved 4 July 2022.
- ^ a b c U.S. Department of Health and Human Services (1 July 2022). "HHS Orders 2.5 Million More Doses of JYNNEOS Vaccine For Monkeypox Preparedness" (Press release). Archived from the original on 4 July 2022. Retrieved 4 July 2022.
- ^ Petersen BW, Damon IK, Pertowski CA, Meaney-Delman D, Guarnizo JT, Beigi RH, et al. (February 2015). "Clinical guidance for smallpox vaccine use in a postevent vaccination program" (PDF). MMWR. Recommendations and Reports. 64 (RR-2): 1–26. PMID 25695372. Archived (PDF) from the original on 19 January 2022. Retrieved 11 August 2022.
- ^ a b c Talbot TR, Stapleton JT, Brady RC, Winokur PL, Bernstein DI, Germanson T, et al. (September 2004). "Vaccination success rate and reaction profile with diluted and undiluted smallpox vaccine: a randomized controlled trial". JAMA. 292 (10): 1205–12. doi:10.1001/jama.292.10.1205. PMID 15353533.
- ^ Costa A (5–7 November 2013). "Smallpox vaccine stockpile" (PDF). World Health Organization (WHO). Archived (PDF) from the original on 4 July 2022. Retrieved 4 July 2022.
- ^ "Smallpox eradication: destruction of variola virus stocks" (PDF). 4 April 2019. Archived (PDF) from the original on 16 May 2022. Retrieved 4 July 2022.
- ^ "Plan national de réponse à une menace de variole" (PDF). Ministère de la Santé et des Solidarités (France). August 2006. Archived (PDF) from the original on 19 May 2022. Retrieved 4 July 2022.
- ^ "Vaiolo delle scimmie, 'abbiamo 5 milioni dosi di vaccino: pronti se servirà'" (in Italian). adnkronos. 27 May 2022. Archived from the original on 14 July 2022. Retrieved 14 July 2022.
- ^ Jihyun Kim (23 May 2022). "15개국 퍼진 원숭이 두창, 불안 확산…백신·치료제는?". Newsis. Seoul. Archived from the original on 23 May 2022. Retrieved 23 May 2022.
- ^ Oh MD, Lee JK (July 2012). "Milestones in history of adult vaccination in Korea". Clinical and Experimental Vaccine Research. 1 (1): 9–17. doi:10.7774/cevr.2012.1.1.9. PMC 3623517. PMID 23596574.
- ^ Ryan KJ, Ray CG, eds. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. pp. 525–28. ISBN 978-0-8385-8529-0.
- ^ a b c Riedel S (January 2005). "Edward Jenner and the history of smallpox and vaccination". Proceedings. 18 (1): 21–25. doi:10.1080/08998280.2005.11928028. PMC 1200696. PMID 16200144.
- ^ Van Alphen J, Aris A (1995). "Medicine in India". Oriental Medicine: An Illustrated Guide to the Asian Arts of Healing. London: Serindia Publications. pp. 19–38. ISBN 978-0-906026-36-6.
- ^ Needham J (1999). "Part 6, Medicine". Science and Civilization in China: Volume 6, Biology and Biological Technology. Cambridge: Cambridge University Press. p. 134.
- ^ Temple R (1986). The Genius of China: 3,000 Years of Science, Discovery, and Invention. New York: Simon and Schuster. p. 137. ISBN 978-0-671-62028-8.
- ^ Silverstein AM (2009). A History of Immunology (2nd ed.). Academic Press. p. 293. ISBN 9780080919461. Archived from the original on 3 August 2020. Retrieved 6 July 2017..
- ^ Voltaire (1742). "Letter XI". Letters on the English. Archived from the original on 16 October 2018. Retrieved 6 July 2017.
- ^ a b Behbehani AM (December 1983). "The smallpox story: life and death of an old disease". Microbiological Reviews. 47 (4): 455–509. doi:10.1128/MMBR.47.4.455-509.1983. PMC 281588. PMID 6319980.
- ^ Aboul-Enein BH, Ross MW, Aboul-Enein FH (2012). "Smallpox inoculation and the Ottoman contribution: A Brief Historiography" (PDF). Texas Public Health Journal. 64 (1): 12. Archived (PDF) from the original on 11 October 2021. Retrieved 23 December 2018.
- ^ Livingstone, N. 2015. The Mistresses of Cliveden. Three centuries of scandal, power and intrigue (p. 229)
- ^ Kennedy P (1715). An Essay on External Remedies Wherein it is Considered, Whether all the curable Distempers incident to Human Bodies, may not be cured by Outward Means. London: A. Bell.
- ^ Willoughby B (12 February 2004). "Black History Month II: Why Wasn't I Taught That?". Tolerance in the News. Archived from the original on 14 January 2009. Retrieved 4 December 2008.
- ^ "Open Collections Program: Contagion, The Boston Smallpox Epidemic, 1721". Archived from the original on 26 July 2018. Retrieved 27 August 2008.
- ^ Robertson P (1974). The book of firsts. New York: C. N. Potter : distributed by Crown Publishers. ISBN 978-0-517-51577-8.
- ^ "Montagu, Turkish Embassy Letters". Archived from the original on 15 April 2013. Retrieved 4 December 2008.
- ^ "Statue of Dr Edward Jenner near the Italian Fountains, Kensington Gardens". lachlan.bluehaze.com.au. Archived from the original on 28 March 2006. Retrieved 16 October 2019.
- ^ Blower S, Bernoulli D (2004). "An attempt at a new analysis of the mortality caused by smallpox and of the advantages of inoculation to prevent it. 1766" (PDF). Reviews in Medical Virology. 14 (5): 275–288. doi:10.1002/rmv.443. PMID 15334536. S2CID 8169180. Archived from the original (PDF) on 27 September 2007.
- ^ Pearson G, ed. (1798). An inquiry concerning the history of the cowpox, principally with a view to supersede and extinguish the smallpox. London, England: J. Johnson. pp. 102–104. Archived from the original on 25 January 2022. Retrieved 31 December 2015.
- ^ Thurston L, Williams G (2015). "An examination of John Fewster's role in the discovery of smallpox vaccination". The Journal of the Royal College of Physicians of Edinburgh. 45 (2): 173–179. doi:10.4997/JRCPE.2015.217. PMID 26181536.
- ^ Michael J. Bennett, War against Smallpox: Edward Jenner and the Global Spread of Vaccination (Cambridge, United Kingdom: Cambridge University Press, 2020), 32.
- ^ a b Bailey I (May 1996). "Edward Jenner (1749–1823): naturalist, scientist, country doctor, benefactor to mankind". Journal of Medical Biography. 4 (2): 63–70. doi:10.1177/096777209600400201. PMID 11616266. S2CID 30318738.
- ^ Hammarsten JF, Tattersall W, Hammarsten JE (1979). "Who discovered smallpox vaccination? Edward Jenner or Benjamin Jesty?". Transactions of the American Clinical and Climatological Association. 90: 44–55. PMC 2279376. PMID 390826.
- ^ Baxby D (January 1999). "Edward Jenner's unpublished cowpox inquiry and the Royal Society: Everard Home's report to Sir Joseph Banks". Medical History. 43 (1): 108–110. doi:10.1017/S0025727300064747. PMC 1044113. PMID 10885136.
- ^ Winkelstein W (1992). "Not just a country doctor: Edward Jenner, scientist". Epidemiologic Reviews. 14: 1–15. doi:10.1093/oxfordjournals.epirev.a036081. PMID 1289108./
- ^ Willis NJ (August 1997). "Edward Jenner and the eradication of smallpox". Scottish Medical Journal. 42 (4): 118–121. doi:10.1177/003693309704200407. PMID 9507590. S2CID 43179073.
- ^ a b Baxby D (January 1999). "Edward Jenner's Inquiry; a bicentenary analysis". Vaccine. 17 (4): 301–307. doi:10.1016/S0264-410X(98)00207-2. PMID 9987167.
- ^ a b c Baxby D (2001). Smallpox Vaccine, Ahead of Its Time – How the Late Development of Laboratory Methods and Other Vaccines Affected the Acceptance of Smallpox Vaccine. Berkeley, UK: Jenner Museum. pp. 12–16. ISBN 978-0-9528695-1-1.
- ^ a b Baxby D (1981). Jenner's smallpox vaccine; the riddle of vaccinia virus and its origin. London: Heinemann Educational Books. ISBN 0-435-54057-2.
- ^ Piercey T (August 2002). "Plaque in Memory of Rev. John Clinch". Archived from the original on 20 March 2018. Retrieved 28 May 2014.
- ^ Handcock G (1996). The Story of Trinity. Trinity: The Trinity Historical Society. p. 1. ISBN 978-098100170-8.
- ^ "First X, Then Y, Now Z : Landmark Thematic Maps – Medicine". Princeton University Library. 2012. Archived from the original on 13 September 2018. Retrieved 22 May 2018.
- ^ Morman ET (2006). "Smallpox". In Finkelman P (ed.). Encyclopedia of the New American Nation. Charles Scribner's Sons. pp. 207–08.
- ^ a b Hopkins DR (2002). The greatest killer : smallpox in history, with a new introduction. Chicago: University of Chicago Press. ISBN 978-0-226-35168-1.
- ^ a b Bazin H (2000). The Eradication of Smallpox. London: Academic Press. pp. 94–102. ISBN 978-0-12-083475-4.
- ^ Rusnock A (2009). "Catching cowpox: the early spread of smallpox vaccination, 1798-1810". Bulletin of the History of Medicine. 83 (1): 17–36. doi:10.1353/bhm.0.0160. PMID 19329840. S2CID 24344691.
- ^ Smith MM (1970). "The 'Real Expedición Marítima de la Vacuna' in New Spain and Guatemala". Trans. Am. Philos. Soc. New Series. 64 (4): 1–74. doi:10.2307/1006158. JSTOR 1006158.
- ^ Ranscombe P (July 2022). "Vaccine Voyages: where science meets slavery". The Lancet Infectious Diseases. 22 (7): 956. doi:10.1016/s1473-3099(22)00270-5. ISSN 1473-3099. PMC 9023002. Archived from the original on 8 July 2022. Retrieved 26 June 2022.
- ^ Tizard IR (2023). A History of Vaccines and Their Opponents. Elsevier. p. 99.
- ^ "Nova et Vetera". British Medical Journal. 1 (2370): 1297–1298. June 1906. doi:10.1136/bmj.1.2370.1297. PMC 2381502. PMID 20762710.
- ^ Meyer C, Reiter S (December 2004). "[Vaccine opponents and sceptics. History, background, arguments, interaction]". Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz (in German). 47 (12): 1182–1188. doi:10.1007/s00103-004-0953-x. PMID 15583889. S2CID 23282373.
- ^ Klein S, Schöneberg I, Krause G (October 2012). "Vom Zwang zur Pockenschutzimpfung zum Nationalen Impfplan". Bundesgesundheitsblatt (in German). 55. pp. 1512–1523. doi:10.25646/1620.
- ^ Jarlert A (2001). Sveriges Kyrkohistoria. Vol. 6. Stockholm. pp. 33–54.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Rheinberger R (1976). "Zum 200. Geburtstag von Landesphysikus Gebhard Schaedler. Ein Liechtensteinischer Artzt als Pionier der Pockenschutzimpfung.". Jahrbuch des Historischen Vereins fur das Furstentum Liechtenstein. Historischer Verein fur das Furstentum Liechtenstein. Vol. 76. pp. 337–343. Archived from the original on 27 October 2021. Retrieved 27 October 2021.
- ^ Pead PJ (December 2003). "Benjamin Jesty: new light in the dawn of vaccination". Lancet. 362 (9401): 2104–2109. doi:10.1016/S0140-6736(03)15111-2. PMID 14697816. S2CID 4254402.
- ^ Plett PC (2006). "[Peter Plett and other discoverers of cowpox vaccination before Edward Jenner]". Sudhoffs Archiv (in German). 90 (2): 219–232. JSTOR 20778029. PMID 17338405.
- ^ a b Williams G (2010). Angel of Death; the story of smallpox. Basingstoke: Palgrave Macmillan. pp. 162–73. ISBN 978-0-230-27471-6.
- ^ a b c Williamson S (2007). The Vaccination Controversy; the rise, reign and decline of compulsory vaccination. Liverpool: Liverpool University Press. ISBN 9781846310867.
- ^ Durbach N (2002). "Class, Gender, and the Conscientious Objector to Vaccination, 1898-1907". Journal of British Studies. 41 (1): 58–83. doi:10.1086/386254. JSTOR 3070762 – via JSTOR.
- ^ George NA (November 1952). "Compulsory smallpox vaccination; the University City, Missouri, case". Public Health Reports. 67 (11): 1135–1138. doi:10.2307/4588305. JSTOR 4588305. PMC 2030845. PMID 12993980.
- ^ "Toward a Twenty-First-Century Jacobson v. Massachusetts" (PDF). Harvard Law Review. 121 (7). The Harvard Law Review Association: 1823–1824. May 2008. Archived (PDF) from the original on 26 October 2014. Retrieved 13 March 2014.
- ^ "A Brief Biography of Dr. Louis T. Wright". North by South: from Charleston to Harlem, the great migration. Archived from the original on 20 October 2017. Retrieved 23 September 2006.
- ^ "Spotlight on Black Inventors, Scientists, and Engineers". Department of Computer Science of Georgetown University. Archived from the original on 7 September 2006. Retrieved 23 September 2006.
- ^ a b Copeman SM (May 1898). "The Milroy Lectures on the Natural History of Vaccina: Delivered at the Royal College of Physicians". British Medical Journal. 1 (1951): 1312–1318. doi:10.1136/bmj.1.1951.1312. PMC 2411485. PMID 20757828.
- ^ a b Didgeon JA (May 1963). "Development of Smallpox Vaccine in England in the Eighteenth and Nineteenth Centuries". British Medical Journal. 1 (5342): 1367–1372. doi:10.1136/bmj.1.5342.1367. PMC 2124036. PMID 20789814.
- ^ Creighton C (1887). The Natural History of Cowpox and Vaccinal Syphilis. London: Cassell.
- ^ Copeman SM (1892). "The Bacteriology of Vaccine Lymph". In Shelley CE (ed.). Transactions of the Seventh International Congress of Hygiene and Demography. Eyre and Spottiswoode. pp. 319–26. Retrieved 14 January 2014.
- ^ Copeman PW (February 1998). "Extinction of the speckled monster celebrated in 1996". Journal of Medical Biography. 6 (1): 39–42. doi:10.1177/096777209800600108. PMID 11619875. S2CID 8918951.
- ^ Dixon CW (1962). Smallpox. London: J. & A. Churchill. pp. 280–81.
- ^ Special Commission (1900). "Report of the Lancet Special Commission on Glycerinated Calf Lymph Vaccines". Lancet. 155 (4000): 1227–36. doi:10.1016/s0140-6736(01)96895-3.
- ^ Collier LH (March 1955). "The development of a stable smallpox vaccine". The Journal of Hygiene. 53 (1): 76–101. doi:10.1017/S002217240000053X. PMC 2217800. PMID 14367805.
- ^ "Professor Leslie Collier". The Telegraph. 22 March 2011. Archived from the original on 12 January 2022. Retrieved 2 May 2013.
- ^ Baxby D (October 2005). "Development of a stable smallpox vaccine: Collier L. J Hyg 1955; 53: 76–101". Epidemiology and Infection. 133 (Suppl. 1): S25–S27. doi:10.1017/S0950268805004280. PMID 24965243.
- ^ Kirkup JR (2006). The Evolution of Surgical Instruments. Novato, California: Norman Publishing. pp. 419–37. ISBN 978-0-930405-86-1.
- ^ a b Rubin BA (May 1980). "A note on the development of the bifurcated needle for smallpox vaccination". WHO Chronicle. 34 (5): 180–181. PMID 7376638.
- ^ Henderson DA (2009). Smallpox; the death of a disease. Amherst, New York: Prometheus Books. pp. 26–27. ISBN 978-1-59102-722-5.
- ^ "Smallpox > Bioterrorism". U.S. Centers for Disease Control and Prevention (CDC). 19 December 2016. Archived from the original on 19 May 2022. Retrieved 21 May 2022.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ Halsell JS, Riddle JR, Atwood JE, Gardner P, Shope R, Poland GA, et al. (June 2003). "Myopericarditis following smallpox vaccination among vaccinia-naive US military personnel". JAMA. 289 (24): 3283–3289. doi:10.1001/jama.289.24.3283. PMID 12824210.
- ^ Strom B, Stratton K, Anason AP, Baciu A (2005). "4. Lessons Learned from the Smallpox Vaccination Program". The Smallpox Vaccination Program: Public Health in an Age of Terrorism. Washington, DC: National Academies Press. pp. 82–97. doi:10.17226/11240. ISBN 0-309-54877-2. Archived from the original on 14 March 2022. Retrieved 21 August 2022.
- ^ Mackenzie D (22 August 2003). "US smallpox vaccination plan grinds to a halt". New Scientist. Archived from the original on 7 February 2022. Retrieved 1 September 2019.
- ^ Public Services and Procurement Canada (21 April 2022). "Third Generation Smallpox Vaccine (6D024-215700/A)". Government of Canada. Archived from the original on 20 June 2022. Retrieved 20 June 2022.
- ^ Osman L (20 May 2022). "Canada considering smallpox vaccine for monkeypox cases, says Dr. Theresa Tam". CTV News. Archived from the original on 20 June 2022. Retrieved 20 June 2022.
- ^ Public Health Agency of Canada (24 May 2022). "Statement from the Minister of Health on Canada's Response to Monkeypox". Government of Canada. Archived from the original on 11 June 2022. Retrieved 26 May 2022.
- ^ Smithson C, Kampman S, Hetman BM, Upton C (2014). "Incongruencies in Vaccinia Virus Phylogenetic Trees". Computation. 2 (4): 182–98. doi:10.3390/computation2040182. hdl:1828/7374.
- ^ Flint J, Racaniello VR, Rall GF, Hatziioannou T, Skalka AM (7 August 2020). Principles of Virology (5th ed.). John Wiley & Sons. ISBN 978-1-68367-033-9. Archived from the original on 8 July 2022. Retrieved 13 June 2022.
- ^ Jenner E (1798). An Inquiry into the Causes and Effects of the Variolæ Vaccinæ. London: Self-published. Archived from the original on 11 July 2022. Retrieved 11 July 2022.
- ^ Esparza J, Schrick L, Damaso CR, Nitsche A (December 2017). "Equination (inoculation of horsepox): An early alternative to vaccination (inoculation of cowpox) and the potential role of horsepox virus in the origin of the smallpox vaccine". Vaccine. 35 (52): 7222–7230. doi:10.1016/j.vaccine.2017.11.003. PMID 29137821.
- ^ Taylor HH (October 1889). "What is Vaccinia?". British Medical Journal. 2 (1504): 951–52. ISSN 0007-1447. PMC 2155820.
- ^ Smith GL, Vanderplasschen A (1998). "Extracellular Enveloped Vaccinia virus: Entry, Egress, and Evasion". In Enjuanes L, Siddel SG, Spaan W (eds.). Coronaviruses and Arteriviruses. Vol. 440. Springer Science & Business Media. p. 396. ISBN 978-0-306-45910-8. PMID 9782308.
- ^ ICTV Taxonomy history: Cowpox virus. 14 April 2021. Archived from the original on 15 April 2021. Retrieved 15 April 2021.
Varidnaviria > Bamfordvirae > Nucleocytoviricota > Pokkesviricetes > Chitovirales > Poxviridae > Chordopoxvirinae > Orthopoxvirus > Cowpox virus
- ^ Tulman ER, Delhon G, Afonso CL, Lu Z, Zsak L, Sandybaev NT, et al. (September 2006). "Genome of horsepox virus". Journal of Virology. 80 (18): 9244–9258. doi:10.1128/JVI.00945-06. PMC 1563943. PMID 16940536.
- ^ Esparza J (September 2013). "Has horsepox become extinct?". The Veterinary Record. 173 (11): 272–273. doi:10.1136/vr.f5587. PMID 24057497. S2CID 36975171.
- ^ a b Duggan AT, Klunk J, Porter AF, Dhody AN, Hicks R, Smith GL, et al. (July 2020). "The origins and genomic diversity of American Civil War Era smallpox vaccine strains". Genome Biology. 21 (1): 175. doi:10.1186/s13059-020-02079-z. PMC 7370420. PMID 32684155.
- ^ Schrick L, Tausch SH, Dabrowski PW, Damaso CR, Esparza J, Nitsche A (October 2017). "An Early American Smallpox Vaccine Based on Horsepox". The New England Journal of Medicine. 377 (15): 1491–1492. doi:10.1056/NEJMc1707600. PMID 29020595.
- ^ Damaso CR (February 2018). "Revisiting Jenner's mysteries, the role of the Beaugency lymph in the evolutionary path of ancient smallpox vaccines". The Lancet Infectious Diseases. 18 (2): e55–e63. doi:10.1016/S1473-3099(17)30445-0. PMID 28827144. Archived from the original on 21 May 2020. Retrieved 13 June 2022.
- ^ a b Brinkmann A, Souza AR, Esparza J, Nitsche A, Damaso CR (December 2020). "Re-assembly of nineteenth-century smallpox vaccine genomes reveals the contemporaneous use of horsepox and horsepox-related viruses in the USA". Genome Biology. 21 (1): 286. doi:10.1186/s13059-020-02202-0. PMC 7716468. PMID 33272280.
- ^ Transactions of the International medical congress, seventh session, held in London, August 2d to 9th, 1881. J. W. Kolckmann. 1881. Archived from the original on 23 May 2022. Retrieved 23 May 2022.
- ^ Esparza J, Lederman S, Nitsche A, Damaso CR (June 2020). "Early smallpox vaccine manufacturing in the United States: Introduction of the "animal vaccine" in 1870, establishment of "vaccine farms", and the beginnings of the vaccine industry". Vaccine. 38 (30): 4773–4779. doi:10.1016/j.vaccine.2020.05.037. PMC 7294234. PMID 32473878.
To honor Jenner, in 1881 Louis Pasteur proposed at the 7th International Congress of Medicine held in London, to generalize the term "vaccination" to refer to all protective immunization procedures against any infectious diseases [19] and since then we talk about vaccines against different diseases.
- ^ Baxby D (February 1999). "Edward Jenner's inquiry after 200 years". BMJ. 318 (7180): 390. doi:10.1136/bmj.318.7180.390. PMC 1114848. PMID 9933209.
Unjustified credit is sometimes given to Jenner for introducing the terms virus (already long used to indicate a transmissible poison) and vaccination (introduced by his friend Richard Dunning in 1800.
Further reading
- Ramsay M, ed. (September 2022) [March 2013]. "Smallpox and mpox (monkeypox): the green book, chapter 29". Immunisation against infectious disease. Public Health England.
External links
- Smallpox U.S. Centers for Disease Control and Prevention
- Smallpox Center for Infectious Disease Research and Policy
- "Smallpox/Monkeypox Vaccine Information Statement (VIS)". U.S. Centers for Disease Control and Prevention (CDC). 19 May 2023.
- "Interim Clinical Considerations for Use of JYNNEOS Vaccine for Mpox Prevention in the United States". U.S. Centers for Disease Control and Prevention (CDC). 26 August 2024.
- "Medication Guide Smallpox (Vaccinia) Vaccine, Live ACAM2000" (PDF). Emergent Biosolutions.

















