Potency and safety analysis of hemp-derived delta-9 products: The hemp vs. cannabis demarcation problem
Contents
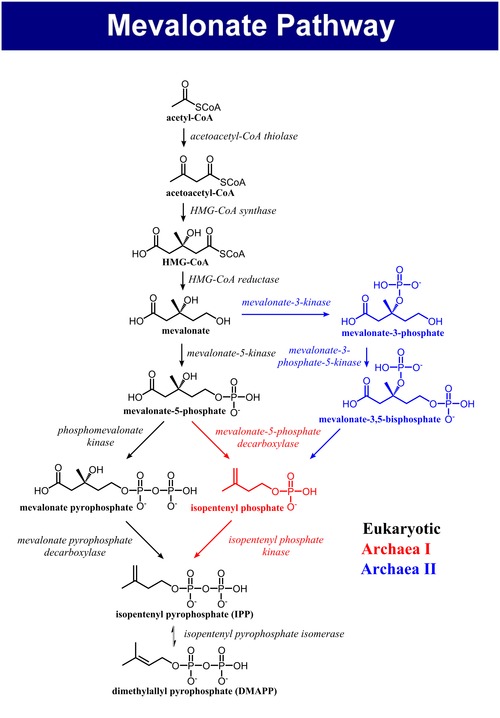
The mevalonate pathway, also known as the isoprenoid pathway or HMG-CoA reductase pathway is an essential metabolic pathway present in eukaryotes, archaea, and some bacteria.[1] The pathway produces two five-carbon building blocks called isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP), which are used to make isoprenoids, a diverse class of over 30,000 biomolecules such as cholesterol, vitamin K, coenzyme Q10, and all steroid hormones.[2]
The mevalonate pathway begins with acetyl-CoA and ends with the production of IPP and DMAPP.[3] It is best known as the target of statins, a class of cholesterol lowering drugs. Statins inhibit HMG-CoA reductase within the mevalonate pathway.
Upper mevalonate pathway
The mevalonate pathway of eukaryotes, archaea, and eubacteria all begin the same way. The sole carbon feed stock of the pathway is acetyl-CoA. The first step condenses two acetyl-CoA molecules to yield acetoacetyl-CoA. This is followed by a second condensation to form HMG-CoA (3-hydroxy-3- methyl-glutaryl-CoA). Reduction of HMG-CoA yields (R)-mevalonate. These first 3 enzymatic steps are called the upper mevalonate pathway.[4]
Lower mevalonate pathway
The lower mevalonate pathway which converts (R)-mevalonate into IPP and DMAPP has 3 variants. In eukaryotes, mevalonate is phosphorylated twice in the 5-OH position, then decarboxylated to yield IPP.[4] In some archaea such as Haloferax volcanii, mevalonate is phosphorylated once in the 5-OH position, decarboxylated to yield isopentenyl phosphate (IP), and finally phosphorylated again to yield IPP (Archaeal Mevalonate Pathway I).[5] A third mevalonate pathway variant found in Thermoplasma acidophilum, phosphorylates mevalonate at the 3-OH position followed by phosphorylation at the 5-OH position. The resulting metabolite, mevalonate-3,5-bisphosphate, is decarboxylated to IP, and finally phosphorylated to yield IPP (Archaeal Mevalonate Pathway II).[6][7]
Regulation and feedback
Several key enzymes can be activated through DNA transcriptional regulation on activation of SREBP (sterol regulatory element-binding protein-1 and -2). This intracellular sensor detects low cholesterol levels and stimulates endogenous production by the HMG-CoA reductase pathway, as well as increasing lipoprotein uptake by up-regulating the LDL-receptor. Regulation of this pathway is also achieved by controlling the rate of translation of the mRNA, degradation of reductase and phosphorylation.[1]
Pharmacology
A number of drugs target the mevalonate pathway:
- Statins (used to decrease cholesterol levels);
- Bisphosphonates (used to treat various bone-degenerative diseases such as osteoporosis[8])
Diseases
A number of diseases affect the mevalonate pathway:
- Mevalonate Kinase Deficiency
- Mevalonic Aciduria
- Hyperimmunoglobulinemia D Syndrome (HIDS).
Alternative pathway
Plants, most bacteria, and some protozoa such as malaria parasites have the ability to produce isoprenoids using an alternative pathway called the methylerythritol phosphate (MEP) or non-mevalonate pathway.[9] The output of both the mevalonate pathway and the MEP pathway are the same, IPP and DMAPP, however the enzymatic reactions to convert acetyl-CoA into IPP are entirely different. Interaction between the two metabolic pathways can be studied by using 13C-glucose isotopomers.[10] In higher plants, the MEP pathway operates in plastids while the mevalonate pathway operates in the cytosol.[9] Examples of bacteria that contain the MEP pathway include Escherichia coli and pathogens such as Mycobacterium tuberculosis.
Enzymatic reactions
| Enzyme | Reaction | Description |
| Acetoacetyl-CoA thiolase |  |
Acetyl-CoA (citric acid cycle) undergoes condensation with another acetyl-CoA molecule to form acetoacetyl-CoA |
| HMG-CoA synthase |  |
Acetoacetyl-CoA condenses with another Acetyl-CoA molecule to form 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA). |
| HMG-CoA reductase |  |
HMG-CoA is reduced to mevalonate by NADPH. This is the rate limiting step in cholesterol synthesis, which is why this enzyme is a good target for pharmaceuticals (statins). |
| mevalonate-5-kinase |  |
Mevalonate is phosphorylated at the 5-OH position to yield mevalonate-5-phosphate (also called phosphomevalonic acid). |
| mevalonate-3-kinase |  |
Mevalonate is phosphorylated at the 3-OH position to yield mevalonate-3-phosphate. 1 ATP is consumed. |
| mevalonate-3-phosphate-5-kinase | 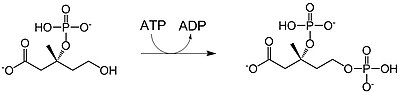 |
Mevalonate-3-phosphate is phosphorylated at the 5-OH position to yield mevalonate-5-phosphate (also called phosphomevalonic acid). 1 ATP is consumed. |
| phosphomevalonate kinase | 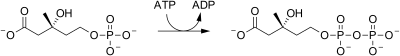 |
mevalonate-5-phosphate is phosphorylated to yield mevalonate-5-pyrophosphate. 1 ATP is consumed. |
| mevalonate-5-pyrophosphate decarboxylase |  |
Mevalonate-5-pyrophosphate is decarboxylated to yield isopentenyl pyrophosphate (IPP). 1 ATP is consumed. |
| isopentenyl pyrophosphate isomerase | 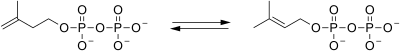 |
isopentenyl pyrophosphate is isomerized to dimethylallyl pyrophosphate. |
References
- ^ a b Buhaescu I, Izzedine H (2007) Mevalonate pathway: areview of clinical and therapeutical implications. ClinBiochem 40:575–584.
- ^ Holstein, S. A., and Hohl, R. J. (2004) Isoprenoids: Remarkable Diversity of Form and Function. Lipids 39, 293−309
- ^ Goldstein, J. L., and Brown, S. B. (1990) Regulation of the mevalonate pathway. Nature 343, 425−430
- ^ a b Miziorko H (2011) Enzymes of the mevalonate pathway of isoprenoid biosynthesis. Arch Biochem Biophys 505:131-143.
- ^ Dellas, N., Thomas, S. T., Manning, G., and Noel, J. P. (2013) Discovery of a metabolic alternative to the classical mevalonate pathway. eLife 2, e00672
- ^ Vinokur JM, Korman TP, Cao Z, Bowie JU (2014) Evidence of a novel mevalonate pathway in archaea. Biochemistry 53:4161–4168.
- ^ Azami Y, Hattori A, Nishimura H, Kawaide H, YoshimuraT, Hemmi H (2014) (R)-mevalonate-3-phosphate is an intermediate of the mevalonate pathway in Thermoplasma acidophilum. J Biol Chem 289:15957–15967.
- ^ Lewiecki, E. Michael (May 2010). "Bisphosphonates for the treatment of osteoporosis: insights for clinicians". Therapeutic Advances in Chronic Disease. 1 (3): 115–128. doi:10.1177/2040622310374783. ISSN 2040-6223. PMC 3513863. PMID 23251734.
- ^ a b Banerjee A, Sharkey TD. (2014) Methylerythritol 4-phosphate (MEP) pathway metabolic regulation. Nat Prod Rep 31:10431055
- ^ Orsi E, Beekwilder J, Peek S, Eggink G, Kengen SW, Weusthuis RA (2020). "Metabolic flux ratio analysis by parallel 13C labeling of isoprenoid biosynthesis in Rhodobacter sphaeroides". Metabolic Engineering. 57: 228–238. doi:10.1016/j.ymben.2019.12.004. PMID 31843486.
External links
- Rensselaer Polytechnic Institute page on cholesterol synthesis (including regulation)


















