Type a search term to find related articles by LIMS subject matter experts gathered from the most trusted and dynamic collaboration tools in the laboratory informatics industry.
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.916 |
| Chemical and physical data | |
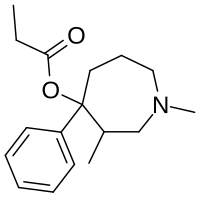
| Formula | C17H25NO2 |
| Molar mass | 275.392 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Proheptazine is an opioid analgesic related to pethidine. It was invented in the 1960s.[2]
Proheptazine produces similar effects to other opioids,[3] including analgesia, sedation, euphoria, dizziness and nausea.
In the United States it is a Schedule I Narcotic controlled substance with an ACSCN of 9643 and a 2013 annual aggregate manufacturing quota of zero. The salts in use are the citrate (free base conversion ratio 0.589), hydrobromide (0.773), and hydrochloride (0.883).[4][5]