Effects of the storage conditions on the stability of natural and synthetic cannabis in biological matrices for forensic toxicology analysis: An update from the literature
Sadržaj
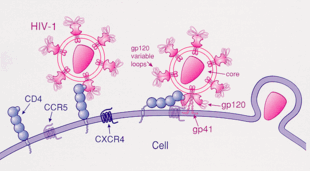
1 – gp120 virusni protein se veže za CD4.
2 – gp120 varijabilna petlja se pričvršćuje na koreceptor, bilo CCR5 ili CXCR4.
3 – HIV ulazi u ćeliju.
C-C hemokinski receptor tip 5, znan i kao CCR5 ili CD195, je protein na površini leukocita, uključen imunski sistem, kao receptor za hemokine.[5]
Kod ljudi, određene populacije naslijedile su mutaciju Delta 32, što je rezultiralo delecijom dijela gena CCR5. Homozigot ni nositelji ove mutacije otporni su na M-tropske sojeve HIV-1 infekcije.[6][7][8][9][10][11]
Funkcija
Protein CCR5 pripada porodici beta hemokinskih receptora, integralnih membranskih proteina.[12][13] To je G-protein spregnuti receptor[12] koji djeluje kao hemokinski receptor u CC hemokinskoj grupi.
CCR5-u srodni ligandi uključuju CCL3, CCL4 (poznat i kao MIP 1α i 1β ) i CCL3L1.[14][15] CCR5 nadalje komunicira sa CCL5 (hemotaksijskim citokinskim proteinom, poznatim i kao RANTES).[14][16][17]
CCR5 je pretežno izražen na T-ćelijama, makrofagima, dendritskim ćelijama, eozinofiliima, mikrogliji i subpopulacijama ćelija karcinoma dojke ili prostate.[18][19] Ekspresija CCR5 selektivno se inducira tokom procesa transformacije raka i ne ispoljava se u normalnim epitelnim ćelijama dojke ili prostate. Otprilike 50% ljudskog karcinoma dojke ispoljilo je CCR5, prvenstveno kod trostruko negativnog karcinoma dojke.[18] CCR5 inhibitors blocked the migration and metastasis of breast and prostate cancer cells that expressed CCR5, suggesting that CCR5 may function as a new therapeutic target.[18][19][20] Nedavna istraživanja sugeriraju da se CCR5 eksprimira u podgrupi ćelija karcinoma sa karakteristikama matičnih ćelija karcinoma, za koje je poznato da podstiču terapijsku rezistenciju, te da su inhibitori CCR5 povećali broj ćelija trenutno ubijenih hemoterapijom.[21] Vjerovatno je da CCR5 ima ulogu u upalnom odgovoru na infekciju, iako njegova tačna uloga u normalnoj imunskoj funkciji nije jasna. Regije ovog proteina su takođe ključne za vezanje hemokin-liganda, kao funkcionalni odgovor receptora i aktivnost koreceptora za HIV.[22]

HIV
HIV-1 najčešće koristi hemokinske receptore CCR5 i/ili CXCR4 kao koreceptor za ulazak u ciljne imunske ćelije.[23] Ovi receptori nalaze se na površini imunskih ćelija domaćina, čime daju mogućnost ulaska virusa HIV-1 da zarazi ćeliju.[24] The HIV-1 envelope glycoprotein structure is essential in enabling the viral entry of HIV-1 into a target host cell.[24] The envelope glycoprotein structure consists of two protein subunits cleaved from a Gp160 protein precursor encoded for by the HIV-1 env gene: the Gp120 external subunit and the Gp41 transmembrane subunit.[24] Ova glikoproteinska struktura omotača raspoređena je u strukturu sličnu klasi koja se nalazi na površini viriona i sastoji se od trimera Gp120-Gp41 heterodimera.[24] The Gp120 envelope protein is a chemokine mimic. Iako mu nedostaje jedinstvena struktura kemokina, i dalje je sposoban da se veže za CCR5 i CXCR4 hemokinske receptore. Tokom HIV-1 infekcije, podjedinica glikoproteinskog omotača Gp120 veže se za glikoprotein CD4 i koreceptor za HIV-1, eksprimiran na ciljnoj ćeliji, formirajući heterotrimerni kompleks. Stvaranje ovog kompleksa stimulira oslobađanje fuzogenog peptida, zbog čega se virusna membrana spaja s membranom ciljanih ćelija domaćina. Budući da vezanje samo za CD4 ponekad može rezultirati oslobađanjem gp120, gp120 se mora dalje vezati za koreceptor CCR5 kako bi fuzija nastavila. Tirozin-sulfatni amino-kraj ovog ko-receptora je "bitna odrednica" vezanja za gp120 glikoprotein.[25] Koreceptor također prepoznaje V1-V2 regiju gp120 i premošćujući sloj (antiparalelni, 4-lančani β sloj koji povezuje unutrašnji i vanjki domen gp120). Drška V1-V2 može uticati na "upotrebu koreceptora putem peptidnog sastava, kao i stupnjem N-vezane glikozilacije". Međutim, za razliku od V1-V2, petlja V3 vrlo je varijabilna i stoga je najvažnija odrednica specifičnosti ko-receptora. Normalni ligandi za ovaj receptor, RANTES, MIP-1β i MIP-1α, sposobni su suzbiti HIV-1 infekciju in vitro. Kod osoba zaraženih HIV-om, virusi koji koriste CCR5 su pretežni tipovi izolirani tokom ranih stadija virusne infekcije,[26] što sugerira da ovi virusi mogu imati selektivnu prednost tokom prenosa ili akutne faze bolesti. Štaviše, najmanje polovina svih zaraženih osoba, u toku infekcije ima samo viruse koji koriste CCR5.
CCR5 je primarni ko-receptor koji gp120 koristi uzastopno sa CD4. Rezultat ovog vezanja je da se gp41, drugi proteinski proizvod gp160, oslobodi iz svoje metastabilne konformacije i ubaci u membranu ćelije domaćina. Iako nije potvrđeno, vezanje gp120-CCR5 uključuje dva ključna koraka: (1) tirozin-sulfatni amino kraj ovog koreceptora je "bitna odrednica" vezanja za gp120 i (2) nakon koraka 1, mora postojati uzajamno djelovanje (sinergija, međusobna komunikacija) između gp120 i CCR5 transmembranskih domena.
CCR5 je neophodan za širenje R5-soja virusa HIV-1 .[27] Poznavanje mehanizma kojim ovaj soj HIV-1 posreduje infekciju podstaknulo je istraživanje u razvoju terapijskih intervencija za blokiranje funkcije CCR5.[28] Brojni novi eksperimentalni HIV lijekovi, zvani CCR5 antagonist receptora, dizajnirani su da ometaju vezanje između proteinskog omotača Gp120 i HIV koreceptora CCR5. Ovi eksperimentalni lijekovi uključuju PRO140 (CytoDyn), Vicriviroc (ispitivanja faze III otkazana su u julu 2010.) (Schering plug, Aplaviroc (GW-873140) (GlaxoSmithKline) i Maraviroc (UK-427857) (Pfizer). FDA je odobrila za upotrebu Maraviroc u avgustu 2007. Za kliničku upotrebu, FDA ga je jedino do sada odobrila, čime je postao prvi inhibitor CCR5. Problem ovog pristupa je tšto, iako je CCR5 glavni koreceptor kojim HIV zaražava ćelije, nije jedini takav koreceptor. Moguće je da će se, pod selekcijskim pritiskom HIV razviti u upotrebu drugog koreceptora. Međutim, ispitivanje virusne rezistencije na AD101, molekulski antagonista CCR5, pokazalo je da rezistentni virusi nisu prešli na drugi koreceptor (CXCR4), ali su ustrajali u upotrebi CCR5. Vezali su se za alternativne domene CCR5 ili na receptor sa većim afinitetom. Međutim, budući da još uvijek postoji još jedan koreceptor, vjerovatno je da nedostatak gena CCR5 ne čini imunost na virus. Virus također i dalje ima pristup CD4. Za razliku od CCR5, koji nije potreban (što dokazuju oni koji žive zdravim životom čak i kad im nedostaje gen kao rezultat mutacije delta32), CD4 je presudan u imunskom odbrambenom sistemu tijela.[29] Čak i bez dostupnosti bilo koreceptora (čak i CCR5), virus i dalje može napasti ćelije, ako bi gp41 prošao kroz izmjenu (uključujući njegov citoplazmatski rep), što je rezultiralo neovisnošću CD4 bez potrebe CCR5 i/ili CXCR4 kao ulaznog puta.[30]
Kancer
Ekspresija CCR5 inducira se u epitelnim ćelijama dojke i prostate, nakon transformacije. Indukcija ekspresije CCR5 promovira ćelijsku invaziju, migraciju i metastaze. Indukcija metastaza pak uključuje usmjeravanje na metastatsko mjesto. Pokazalo se da CCR5 inhibitori, uključujući Maraviroc i Leronlimab, blokiraju metastazu na plućnim ćelijskim linijaama karcinoma dojke. U pretkliničkim ispitivanjima imunih miševa, CCR5 inhibitori blokirali su metastaze u kostima i mozgu. CCR5 inhibitori također smanjuju infiltraciju makrofaga povezanih s tumorom.[31] Studija faze 1 kliničkog ispitivanja inhibitora CCR5 kod teško liječenih pacijenata sa metastatskim karcinomom debelog crijeva pokazala je objektivan klinički odgovor i smanjenje metastatskog opterećenja tumora.[32]
Mozak
Povećani nivoi CCR5 dio su upalnog odgovora na moždani udar. Blokiranje CCR5 lijekom Maraviroc (lijek odobren za HIV) može poboljšati oporavak nakon udara.[33][34]
U mozgu u razvoju, hemokini poput CCR5 utiču na migracije i veze neurona. Nakon moždanog udara, čini se da smanjuju broj mjesta povezivanja na neuronima u blizini oštećenja.
CCR5-Δ32
CCR5-Δ32 (ili CCR5-D32 ili CCR5 delta 32) je alel CCR5-a.
CCR5-Δ32
CCR5-Δ32 (ili CCR5-D32 ili CCR5 delta 32) je alel CCR5-a.[35][36]
CCR5 Δ32 je delecija u paru od 32 baze, koja uvodi prerano stop kodon u CCR5 receptorski lokus, što rezultira nefunkcionalnim receptorom.[37][38] CCR5 is required for M-tropic HIV-1 virus entry.[39] Homozigotne osobe (označeno sa Δ32 / Δ32) za CCR5 Δ32 ne ispoljavaju funkcionalne CCR5 receptore na ćelijskim površinama i otporni su na infekciju HIV-1-om, uprkos višestrukoj izloženosti visokom riziku. Heterozigotne (+ / Δ32) za mutantni alel imaju više od 50% smanjenje funkcionalnih CCR5 receptora na površinama ćelija, zbog dimerizacije između mutiranih i divljih receptora, koji ometaju transportom CCR5 na površinu ćelije.[40] Heterozygote carriers are resistant to HIV-1 infection relative to wild types and when infected, heterozygotes exhibit reduced viral loads and a 2-3-year-slower progression to AIDS relative to wild types.[37][39][41] Heterozigoznost za ovaj mutirani alel također je pokazala da poboljšava odgovor na virus, na antiretrovirusni tretman.[42] U Evropi, CCR5 Δ32 ima učestalost alela (heterozigota) od 10%, a frekvenciju homozigota od 1%.
Nedavna istraživanja pokazuju da CCR5 Δ32 poboljšava kogniciju i pamćenje. U 2016. istraživanja su pokazala da je uklanjanje gena CCR5 miševa značajno poboljšalo njihovo pamćenje.[43] CCR5 is a powerful suppressor for neuronal plasticity, learning, and memory; CCR5 over-activation by viral proteins may contribute to HIV-associated cognitive deficits.[44]
Također pogledajte
Reference
- ^ a b c GRCh38: Ensembl release 89: ENSG00000160791 - Ensembl, maj 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000079227 - Ensembl, maj 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Jiao X, Nawab O, Patel T, Kossenkov AV, Halama N, Jaeger D, Pestell RG (Oct 2019). "Recent Advances Targeting CCR5 for Cancer and Its Role in Immuno-Oncology". Cancer Research. 79 (19): 4801–4807. doi:10.1158/0008-5472.CAN-19-1167. PMC 6810651. PMID 31292161.
- ^ de Silva E, Stumpf MP (Dec 2004). "HIV and the CCR5-Delta32 resistance allele". FEMS Microbiology Letters. 241 (1): 1–12. doi:10.1016/j.femsle.2004.09.040. PMID 15556703.
- ^ Hütter G, Nowak D, Mossner M, Ganepola S, Müssig A, Allers K, Schneider T, Hofmann J, Kücherer C, Blau O, Blau IW, Hofmann WK, Thiel E (Feb 2009). "Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation". The New England Journal of Medicine. 360 (7): 692–8. doi:10.1056/NEJMoa0802905. PMID 19213682.
- ^ Allers K, Hütter G, Hofmann J, Loddenkemper C, Rieger K, Thiel E, Schneider T (Mar 2011). "Evidence for the cure of HIV infection by CCR5Δ32/Δ32 stem cell transplantation". Blood. 117 (10): 2791–9. doi:10.1182/blood-2010-09-309591. PMID 21148083.
- ^ Zhen A, Kitchen S (Jan 2014). "Stem-cell-based gene therapy for HIV infection". Viruses. 6 (1): 1–12. doi:10.3390/v6010001. PMC 3917429. PMID 24368413.
- ^ Kay MA, Walker BD (Mar 2014). "Engineering cellular resistance to HIV". The New England Journal of Medicine. 370 (10): 968–9. doi:10.1056/NEJMe1400593. PMID 24597871.
- ^ Tebas P, Stein D, Tang WW, Frank I, Wang SQ, Lee G, et al. (Mar 2014). "Gene editing of CCR5 in autologous CD4 T cells of persons infected with HIV". The New England Journal of Medicine. 370 (10): 901–10. doi:10.1056/NEJMoa1300662. PMC 4084652. PMID 24597865.
- ^ a b "CCR5 - chemokine (C-C motif) receptor 5 (gene/pseudogene)". Genetics Home Reference. Arhivirano s originala, 24. 9. 2009. Pristupljeno 16. 3. 2021.
- ^ Samson M, Labbe O, Mollereau C, Vassart G, Parmentier M (Mar 1996). "Molecular cloning and functional expression of a new human CC-chemokine receptor gene". Biochemistry. 35 (11): 3362–7. doi:10.1021/bi952950g. PMID 8639485.
- ^ a b Struyf S, Menten P, Lenaerts JP, Put W, D'Haese A, De Clercq E, Schols D, Proost P, Van Damme J (Jul 2001). "Diverging binding capacities of natural LD78beta isoforms of macrophage inflammatory protein-1alpha to the CC chemokine receptors 1, 3 and 5 affect their anti-HIV-1 activity and chemotactic potencies for neutrophils and eosinophils". European Journal of Immunology. 31 (7): 2170–8. doi:10.1002/1521-4141(200107)31:7<2170::AID-IMMU2170>3.0.CO;2-D. PMID 11449371.
- ^ Miyakawa T, Obaru K, Maeda K, Harada S, Mitsuya H (Feb 2002). "Identification of amino acid residues critical for LD78beta, a variant of human macrophage inflammatory protein-1alpha, binding to CCR5 and inhibition of R5 human immunodeficiency virus type 1 replication". The Journal of Biological Chemistry. 277 (7): 4649–55. doi:10.1074/jbc.M109198200. PMID 11734558.
- ^ Slimani H, Charnaux N, Mbemba E, Saffar L, Vassy R, Vita C, Gattegno L (Oct 2003). "Interaction of RANTES with syndecan-1 and syndecan-4 expressed by human primary macrophages". Biochimica et Biophysica Acta (BBA) - Biomembranes. 1617 (1–2): 80–8. doi:10.1016/j.bbamem.2003.09.006. PMID 14637022.
- ^ Proudfoot AE, Fritchley S, Borlat F, Shaw JP, Vilbois F, Zwahlen C, Trkola A, Marchant D, Clapham PR, Wells TN (Apr 2001). "The BBXB motif of RANTES is the principal site for heparin binding and controls receptor selectivity". The Journal of Biological Chemistry. 276 (14): 10620–6. doi:10.1074/jbc.M010867200. PMID 11116158.
- ^ a b c Velasco-Velázquez M, Jiao X, De La Fuente M, Pestell TG, Ertel A, Lisanti MP, Pestell RG (Aug 2012). "CCR5 antagonist blocks metastasis of basal breast cancer cells". Cancer Research. 72 (15): 3839–50. doi:10.1158/0008-5472.CAN-11-3917. PMID 22637726.
- ^ a b Sicoli D, Jiao X, Ju X, Velasco-Velazquez M, Ertel A, Addya S, Li Z, Andò S, Fatatis A, Paudyal B, Cristofanilli M, Thakur ML, Lisanti MP, Pestell RG (Dec 2014). "CCR5 receptor antagonists block metastasis to bone of v-Src oncogene-transformed metastatic prostate cancer cell lines". Cancer Research. 74 (23): 7103–14. doi:10.1158/0008-5472.CAN-14-0612. PMC 4294544. PMID 25452256.
- ^ Velasco-Velázquez M, Xolalpa W, Pestell RG (Nov 2014). "CCL5/CCR5 in breast cancer". Expert Opin Ther Targets. 18 (11): 1265–75. doi:10.1517/14728222.2014.949238. PMID 25256399.
- ^ Jiao X, Velasco-Velázquez MA, Wang M, Li Z, Rui H, Peck AR, Korkola JE, Chen X, Xu S, DuHadaway JB, Guerrero-Rodriguez S, Addya S, Sicoli D, Mu Z, Zhang G, Stucky A, Zhang X, Cristofanilli M, Fatatis A, Gray JW, Zhong JF, Prendergast GC, Pestell RG (Apr 2018). "CCR5 Governs DNA Damage Repair and Breast Cancer Stem Cell Expansion". Cancer Res. 78 (7): 1657–71. doi:10.1158/0008-5472.CAN-17-0915. PMC 6331183. PMID 29358169.
- ^ Barmania F, Pepper MS (2013). "C-C chemokine receptor type five (CCR5): An emerging target for the control of HIV infection". Applied & Translational Genomics. 2 (a): 3–16. doi:10.1016/j.atg.2013.05.004. PMC 5133339. PMID 27942440.
- ^ Murphy PM (Feb 2001). "Viral exploitation and subversion of the immune system through chemokine mimicry". Nature Immunology. 2 (2): 116–22. doi:10.1038/84214. PMID 11175803.
- ^ a b c d Alkhatib G (Mar 2009). "The biology of CCR5 and CXCR4". Current Opinion in HIV and AIDS. 4 (2): 96–103. doi:10.1097/COH.0b013e328324bbec. PMC 2718543. PMID 19339947.
- ^ "CCR5 Inhibitors and HIV".
- ^ Anderson J, Akkina R (Sep 2007). "Complete knockdown of CCR5 by lentiviral vector-expressed siRNAs and protection of transgenic macrophages against HIV-1 infection". Gene Therapy. 14 (17): 1287–97. doi:10.1038/sj.gt.3302958. PMID 17597795.
- ^ Lieberman-Blum SS, Fung HB, Bandres JC (Jul 2008). "Maraviroc: a CCR5-receptor antagonist for the treatment of HIV-1 infection". Clinical Therapeutics. 30 (7): 1228–50. doi:10.1016/s0149-2918(08)80048-3. PMID 18691983.
- ^ Nazari R, Joshi S (Aug 2008). "CCR5 as target for HIV-1 gene therapy". Current Gene Therapy. 8 (4): 264–72. doi:10.2174/156652308785160674. PMID 18691022.
- ^ "Understanding Genetics". The Tech Museum of Innovation, San Jose, CA. Arhivirano s originala, 17. 6. 2021. Pristupljeno 16. 3. 2021.
- ^ Taylor BM, Foulke JS, Flinko R, Heredia A, DeVico A, Reitz M (Jun 2008). "An alteration of human immunodeficiency virus gp41 leads to reduced CCR5 dependence and CD4 independence". Journal of Virology. 82 (11): 5460–71. doi:10.1128/JVI.01049-07. PMC 2395218. PMID 18353949.
- ^ Frankenberger C, Rabe D, Bainer R, Sankarasharma D, Chada K, Krausz T, Gilad Y, Becker L, Rosner MR (Oct 2015). "Metastasis Suppressors Regulate the Tumor Microenvironment by Blocking Recruitment of Prometastatic Tumor-Associated Macrophages". Cancer Res. 75 (19): 4063–73. doi:10.1158/0008-5472.CAN-14-3394. PMC 4592465. PMID 26238785.
- ^ Halama N, Zoernig I, Berthel A, Kahlert C, Klupp F, Suarez-Carmona M, Suetterlin T, Brand K, Krauss J, Lasitschka F, Lerchl T, Luckner-Minden C, Ulrich A, Koch M, Weitz J, Schneider M, Buechler MW, Zitvogel L, Herrmann T, Benner A, Kunz C, Luecke S, Springfeld C, Grabe N, Falk CS, Jaeger D (Apr 2016). "Tumoral Immune Cell Exploitation in Colorectal Cancer Metastases Can Be Targeted Effectively by Anti-CCR5 Therapy in Cancer Patients". Cancer Cell. 29 (4): 587–601. doi:10.1016/j.ccell.2016.03.005. PMID 27070705.
- ^ "HIV drug could improve recovery after stroke". ScienceMag. 21. 2. 2019. Pristupljeno 22. 2. 2019.
- ^ Joy MT, Assayag EB, Shabashov-Stone D, Liraz-Zaltsman S, Mazzitelli J, Arenas M, Abduljawad N, Kliper E, Korczyn AD, Thareja NS, Kesner EL, Zhou M, Huang S, Silva TK, Katz N, Bornstein NM, Silva AJ, Shohami E, Carmichael ST (21. 2. 2019). "CCR5 Is a Therapeutic Target for Recovery after Stroke and Traumatic Brain Injury". Cell. 176 (5): 1143–1157. doi:10.1016/j.cell.2019.01.044. PMC 7259116. PMID 30794775.
- ^ Galvani AP, Slatkin M (Dec 2003). "Evaluating plague and smallpox as historical selective pressures for the CCR5-Delta 32 HIV-resistance allele". Proceedings of the National Academy of Sciences of the United States of America. 100 (25): 15276–9. Bibcode:2003PNAS..10015276G. doi:10.1073/pnas.2435085100. PMC 299980. PMID 14645720.
- ^ Stephens JC, Reich DE, Goldstein DB, Shin HD, Smith MW, Carrington M, et al. (Jun 1998). "Dating the origin of the CCR5-Delta32 AIDS-resistance allele by the coalescence of haplotypes". American Journal of Human Genetics. 62 (6): 1507–15. doi:10.1086/301867. PMC 1377146. PMID 9585595.
- ^ a b Dean M, Carrington M, Winkler C, Huttley GA, Smith MW, Allikmets R, et al. (Sep 1996). "Genetic restriction of HIV-1 infection and progression to AIDS by a deletion allele of the CKR5 structural gene. Hemophilia Growth and Development Study, Multicenter AIDS Cohort Study, Multicenter Hemophilia Cohort Study, San Francisco City Cohort, ALIVE Study". Science. 273 (5283): 1856–62. Bibcode:1996Sci...273.1856D. doi:10.1126/science.273.5283.1856. PMID 8791590.
- ^ Samson M, Libert F, Doranz BJ, Rucker J, Liesnard C, Farber CM, Saragosti S, Lapoumeroulie C, Cognaux J, Forceille C, Muyldermans G, Verhofstede C, Burtonboy G, Georges M, Imai T, Rana S, Yi Y, Smyth RJ, Collman RG, Doms RW, Vassart G, Parmentier M (Aug 1996). "Resistance to HIV-1 infection in caucasian individuals bearing mutant alleles of the CCR-5 chemokine receptor gene". Nature. 382 (6593): 722–5. Bibcode:1996Natur.382..722S. doi:10.1038/382722a0. PMID 8751444.
- ^ a b Liu R, Paxton WA, Choe S, Ceradini D, Martin SR, Horuk R, MacDonald ME, Stuhlmann H, Koup RA, Landau NR (Aug 1996). "Homozygous defect in HIV-1 coreceptor accounts for resistance of some multiply-exposed individuals to HIV-1 infection". Cell. 86 (3): 367–77. doi:10.1016/s0092-8674(00)80110-5. PMID 8756719.
- ^ Benkirane M, Jin DY, Chun RF, Koup RA, Jeang KT (Dec 1997). "Mechanism of transdominant inhibition of CCR5-mediated HIV-1 infection by ccr5delta32". The Journal of Biological Chemistry. 272 (49): 30603–6. doi:10.1074/jbc.272.49.30603. PMID 9388191.
- ^ Michael NL, Louie LG, Rohrbaugh AL, Schultz KA, Dayhoff DE, Wang CE, Sheppard HW (Oct 1997). "The role of CCR5 and CCR2 polymorphisms in HIV-1 transmission and disease progression". Nature Medicine. 3 (10): 1160–2. doi:10.1038/nm1097-1160. PMID 9334732.
- ^ Laurichesse JJ, Persoz A, Theodorou I, Rouzioux C, Delfraissy JF, Meyer L (maj 2007). "Improved virological response to highly active antiretroviral therapy in HIV-1-infected patients carrying the CCR5 Delta32 deletion". HIV Medicine. 8 (4): 213–9. doi:10.1111/j.1468-1293.2007.00455.x. PMID 17461848.
- ^ "China's CRISPR twins might have had their brains inadvertently enhanced". Technology Review. Pristupljeno 22. 2. 2019.
- ^ Zhou M, Greenhill S, Huang S, Silva TK, Sano Y, Wu S, Cai Y, Nagaoka Y, Sehgal M, Cai DJ, Lee YS, Fox K, Silva AJ (20. 12. 2016). "CCR5 is a suppressor for cortical plasticity and hippocampal learning and memory". eLife. 5. doi:10.7554/eLife.20985. PMC 5213777. PMID 27996938.
Dopunska literatura
- Wilkinson D (Sep 1996). "Cofactors provide the entry keys. HIV-1". Current Biology. 6 (9): 1051–3. doi:10.1016/S0960-9822(02)70661-1. PMID 8805353.
- Broder CC, Dimitrov DS (1996). "HIV and the 7-transmembrane domain receptors". Pathobiology. 64 (4): 171–9. doi:10.1159/000164032. PMID 9031325.
- Choe H, Martin KA, Farzan M, Sodroski J, Gerard NP, Gerard C (Jun 1998). "Structural interactions between chemokine receptors, gp120 Env and CD4". Seminars in Immunology. 10 (3): 249–57. doi:10.1006/smim.1998.0127. PMID 9653051.
- Sheppard HW, Celum C, Michael NL, O'Brien S, Dean M, Carrington M, Dondero D, Buchbinder SP (Mar 2002). "HIV-1 infection in individuals with the CCR5-Delta32/Delta32 genotype: acquisition of syncytium-inducing virus at seroconversion". Journal of Acquired Immune Deficiency Syndromes. 29 (3): 307–13. doi:10.1097/00042560-200203010-00013. PMID 11873082.
- Freedman BD, Liu QH, Del Corno M, Collman RG (2003). "HIV-1 gp120 chemokine receptor-mediated signaling in human macrophages". Immunologic Research. 27 (2–3): 261–76. doi:10.1385/IR:27:2-3:261. PMID 12857973.
- Esté JA (Sep 2003). "Virus entry as a target for anti-HIV intervention". Current Medicinal Chemistry. 10 (17): 1617–32. doi:10.2174/0929867033457098. PMID 12871111.
- Gallo SA, Finnegan CM, Viard M, Raviv Y, Dimitrov A, Rawat SS, Puri A, Durell S, Blumenthal R (Jul 2003). "The HIV Env-mediated fusion reaction". Biochimica et Biophysica Acta (BBA) - Biomembranes. 1614 (1): 36–50. doi:10.1016/S0005-2736(03)00161-5. PMID 12873764.
- Zaitseva M, Peden K, Golding H (Jul 2003). "HIV coreceptors: role of structure, posttranslational modifications, and internalization in viral-cell fusion and as targets for entry inhibitors". Biochimica et Biophysica Acta (BBA) - Biomembranes. 1614 (1): 51–61. doi:10.1016/S0005-2736(03)00162-7. PMID 12873765.
- Lee C, Liu QH, Tomkowicz B, Yi Y, Freedman BD, Collman RG (Nov 2003). "Macrophage activation through CCR5- and CXCR4-mediated gp120-elicited signaling pathways". Journal of Leukocyte Biology. 74 (5): 676–82. doi:10.1189/jlb.0503206. PMID 12960231.
- Yi Y, Lee C, Liu QH, Freedman BD, Collman RG (2004). "Chemokine receptor utilization and macrophage signaling by human immunodeficiency virus type 1 gp120: Implications for neuropathogenesis". Journal of Neurovirology. 10 (Suppl 1): 91–6. doi:10.1080/753312758. PMID 14982745.
- Seibert C, Sakmar TP (2004). "Small-molecule antagonists of CCR5 and CXCR4: a promising new class of anti-HIV-1 drugs". Current Pharmaceutical Design. 10 (17): 2041–62. doi:10.2174/1381612043384312. PMID 15279544.
- Cutler CW, Jotwani R (2006). "Oral mucosal expression of HIV-1 receptors, co-receptors, and alpha-defensins: tableau of resistance or susceptibility to HIV infection?". Advances in Dental Research. 19 (1): 49–51. doi:10.1177/154407370601900110. PMC 3750741. PMID 16672549.
- Ajuebor MN, Carey JA, Swain MG (Aug 2006). "CCR5 in T cell-mediated liver diseases: what's going on?". Journal of Immunology. 177 (4): 2039–45. doi:10.4049/jimmunol.177.4.2039. PMID 16887960.
- Lipp M, Müller G (2003). "Shaping up adaptive immunity: the impact of CCR7 and CXCR5 on lymphocyte trafficking". Verhandlungen der Deutschen Gesellschaft für Pathologie. 87: 90–101. PMID 16888899.
- Balistreri CR, Caruso C, Grimaldi MP, Listì F, Vasto S, Orlando V, Campagna AM, Lio D, Candore G (Apr 2007). "CCR5 receptor: biologic and genetic implications in age-related diseases". Annals of the New York Academy of Sciences. 1100 (1): 162–72. Bibcode:2007NYASA1100..162B. doi:10.1196/annals.1395.014. PMID 17460174.
- Madsen HO, Poulsen K, Dahl O, Clark BF, Hjorth JP (Mar 1990). "Retropseudogenes constitute the major part of the human elongation factor 1 alpha gene family". Nucleic Acids Research. 18 (6): 1513–6. doi:10.1093/nar/18.6.1513. PMC 330519. PMID 2183196.
- Uetsuki T, Naito A, Nagata S, Kaziro Y (Apr 1989). "Isolation and characterization of the human chromosomal gene for polypeptide chain elongation factor-1 alpha". The Journal of Biological Chemistry. 264 (10): 5791–8. PMID 2564392.
- Whiteheart SW, Shenbagamurthi P, Chen L, Cotter RJ, Hart GW (Aug 1989). "Murine elongation factor 1 alpha (EF-1 alpha) is posttranslationally modified by novel amide-linked ethanolamine-phosphoglycerol moieties. Addition of ethanolamine-phosphoglycerol to specific glutamic acid residues on EF-1 alpha". The Journal of Biological Chemistry. 264 (24): 14334–41. PMID 2569467.
- Ann DK, Wu MM, Huang T, Carlson DM, Wu R (Mar 1988). "Retinol-regulated gene expression in human tracheobronchial epithelial cells. Enhanced expression of elongation factor EF-1 alpha". The Journal of Biological Chemistry. 263 (8): 3546–9. PMID 3346208.
- Brands JH, Maassen JA, van Hemert FJ, Amons R, Möller W (Feb 1986). "The primary structure of the alpha subunit of human elongation factor 1. Structural aspects of guanine-nucleotide-binding sites". European Journal of Biochemistry / FEBS. 155 (1): 167–71. doi:10.1111/j.1432-1033.1986.tb09472.x. PMID 3512269.
Vanjski linkovi
- CCR5 - C-C motif chemokine receptor 5 (gene/pseudogene) - Homo sapiens (human) Information from NCBI Gene Database
- Crystal Structure of the CCR5 Chemokine Receptor A rotatable, zoomable 3-D image from rcsb.org
- Video and text from a PBS documentary about the discovery of CCR5
- "Chemokine Receptors: CCR5". IUPHAR Database of Receptors and Ion Channels. International Union of Basic and Clinical Pharmacology. Arhivirano s originala, 18. 1. 2021. Pristupljeno 16. 3. 2021.
- HuGENavigator literature on HIV Infections and CCR5 from Centers for Disease Control and Prevention - (note, authors may not be CDC employees, and there is no public domain notice on the page, so this cannot be assumed to be public domain)
- Schering-Plough Initiates Phase III Studies with CCR5-Vicriviroc in Treatment- Experienced HIV Patients.
- HIVcoPred A server for prediction of HIV coreceptor usage (CCR5). PLoS ONE 8(4): e61437
- Lokacija ljudskog genoma CCR5 i stranica sa detaljima o genu CCR5 u UCSC Genome Browseru.
- P51681






















