Does cannabis extract obtained from cannabis flowers with maximum allowed residual level of aflatoxins and ochratoxin A have an impact on human safety and health?
Contents
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Tolvon, others |
| Other names | Mianserin hydrochloride; Org GB 94[1][2] |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 20–30%[4] |
| Protein binding | 95%[4] |
| Metabolism | Liver (CYP2D6; via aromatic hydroxylation, N-oxidation, N-demethylation)[4] |
| Elimination half-life | 21–61 hours[5] |
| Excretion | Urine: 4–7%[4] Feces: 14–28%[4] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.041.884 |
| Chemical and physical data | |
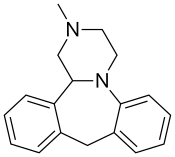
| Formula | C18H20N2 |
| Molar mass | 264.372 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Mianserin, sold under the brand name Tolvon among others, is an atypical antidepressant that is used primarily in the treatment of depression in Europe and elsewhere in the world.[6] It is a tetracyclic antidepressant (TeCA). Mianserin is closely related to mirtazapine, both chemically and in terms of its actions and effects, although there are significant differences between the two drugs (for example, its higher noradrenergic activity and lower 5-HT3 receptor antagonism).[7]
Medical uses
Mianserin at higher doses (30–90 mg/day) is used for the treatment of major depressive disorder.[6]
It can also be used at lower doses (around 10 mg/day) to treat insomnia.[8][9]
Contraindications
It should not be given, except if based on clinical need and under strict medical supervision, to people younger than 18 years old, as it can increase the risk of suicide attempts and suicidal thinking, and it can increase aggressiveness.[6]
While there is no evidence that it can harm a fetus from animal models, there are no data showing it safe for pregnant women to take.[6]
People with severe liver disease should not take mianserin, and it should be used with caution for people with epilepsy or who are at risk for seizures, as it can lower the threshold for seizures. If based on clinical decision, normal precautions should be exercised and the dosages of mianserin and any concurrent therapy kept under review and adjusted as needed.[6]
Side effects
Very common (incidence > 10%) adverse effects include constipation, dry mouth, and drowsiness at the beginning of treatment.[5][6]
Common (1% < incidence ≤ 10%) adverse effects include drowsiness during maintenance therapy, tremor, headache, dizziness, vertigo, and weakness.[5]
Uncommon (0.1% < incidence ≤ 1%) adverse effects include weight gain.[5]
Withdrawal
Abrupt or rapid discontinuation of mianserin may provoke a withdrawal, the effects of which may include depression, anxiety, panic attacks,[10] decreased appetite or anorexia, insomnia, diarrhea, nausea and vomiting, and flu-like symptoms, such as allergies or pruritus, among others.
Overdose
Overdose of mianserin is known to produce sedation, coma, hypotension or hypertension, tachycardia, and QT interval prolongation.[11]
Interactions
Mianserin may enhance the sedative effects of drugs such as alcohol, anxiolytics, hypnotics, or antipsychotics when co-administered. It may decrease the efficacy of antiepileptic medications.
Carbamazepine and phenobarbital will cause the body to metabolize mianserin faster and may reduce its effects. There is a risk of dangerously low blood pressure if people take mianserin along with diazoxide, hydralazine, or nitroprusside. Mianserin can make antihistamines and antimuscarinics have stronger effects. Mianserin should not be taken with apraclonidine, brimonidine, sibutramine, or the combination drug of artemether with lumefantrine.[6]
Pharmacology
Pharmacodynamics
| Site | Ki (nM) | Species | Ref |
|---|---|---|---|
| SERT | 4,000 | Human | [13] |
| NET | 71 | Human | [13] |
| DAT | 9,400 | Human | [13] |
| 5-HT1A | 400–2,600 | Human | [14][15] |
| 5-HT1B | ≥2,800 | Rat | [16] |
| 5-HT1D | 220–400 | Human | [17][18] |
| 5-HT1E | ND | ND | ND |
| 5-HT1F | 13 | Human | [14] |
| 5-HT2A | 1.6–55 | Human | [19][20] |
| 5-HT2B | 1.6–20 | Human | [21][22] |
| 5-HT2C | 0.63–6.5 | Human | [19][23] |
| 5-HT3 | 5.8–300 | Rodent | [24][15] |
| 5-HT4 | ND | ND | ND |
| 5-HT5A | ND | ND | ND |
| 5-HT6 | 55–81 | Human | [25][26] |
| 5-HT7 | 48–56 | Human | [27][28][29] |
| α1 | 34 | Human | [30] |
| α2 | 73 | Human | [30] |
| α2A | 4.8 | Human | [27] |
| α2B | 27 | Human | [31] |
| α2C | 3.8 | Human | [27] |
| D1 | 426–1,420 | Human | [15][27] |
| D2 | 2,100–2,700 | Human | [30][32] |
| D3 | 2,840 | Human | [30] |
| D4 | ND | ND | ND |
| D5 | ND | ND | ND |
| H1 | 0.30–1.7 | Human | [33][30][27] |
| H2 | 437 | Human | [34] |
| H3 | 95,500 | Human | [34] |
| H4 | >100,000 | Human | [34][35] |
| mACh | 820 | Human | [30] |
| MOR | 21,000 | Human | [36] |
| DOR | 30,200 | Human | [36] |
| KOR | 530 (EC50) | Human | [36] |
| Values are Ki (nM), unless otherwise noted. The smaller the value, the more strongly the drug binds to the site. | |||
Mianserin appears to exert its effects via antagonism of histamine and serotonin receptors, and inhibition of norepinephrine reuptake. More specifically, it is an antagonist/inverse agonist at most or all sites of the histamine H1 receptor, serotonin 5-HT1D, 5-HT1F, 5-HT2A, 5-HT2B, 5-HT2C, 5-HT3, 5-HT6, and 5-HT7 receptors, and adrenergic α1- and α2-adrenergic receptors, and additionally a norepinephrine reuptake inhibitor.[37][38] As an H1 receptor inverse agonist with high affinity, mianserin has strong antihistamine effects (e.g., sedation). Conversely, it has low affinity for the muscarinic acetylcholine receptors, and hence lacks anticholinergic properties.[30] Mianserin has been found to be a low affinity but potentially significant partial agonist of the κ-opioid receptor (Ki = 1.7 μM; EC50 = 0.53 μM),[36] similarly to some tricyclic antidepressants (TCAs).[39]
Blockade of the H1 and possibly α1-adrenergic receptors has sedative effects,[5] and also antagonism of the 5-HT2A and α1-adrenergic receptors inhibits activation of intracellular phospholipase C (PLC), which seems to be a common target for several different classes of antidepressants.[40] By antagonizing the somatodendritic and presynaptic α2-adrenergic receptors, which function predominantly as inhibitory autoreceptors and heteroreceptors, mianserin disinhibits the release of norepinephrine, dopamine, serotonin, and acetylcholine in various areas of the brain and body.
Along with mirtazapine, although to a lesser extent in comparison, mianserin has sometimes been described as a noradrenergic and specific serotonergic antidepressant (NaSSA).[41] However, the actual evidence in support of this label has been regarded as poor.[42]
Pharmacokinetics
The bioavailability of mianserin is 20 to 30%.[4] Its plasma protein binding is 95%.[4] Mianserin is metabolized in the liver by the CYP2D6 enzyme via N-oxidation and N-demethylation.[4] Its elimination half-life is 21 to 61 hours.[4] The drug is excreted 4 to 7% in the urine and 14 to 28% in feces.[4]
Chemistry

Mianserin is a tetracyclic piperazinoazepine. Mirtazapine was developed by the same team of organic chemists and differs via addition of a nitrogen atom in one of the rings.[43][44] (S)-(+)-Mianserin is approximately 200–300 times more active than its enantiomer (R)-(−)-mianserin; hence, the activity of mianserin lies in the (S)-(+) isomer.[citation needed]
History
It was developed but not discovered by Organon International; the first patents were issued in The Netherlands in 1967, and it was launched in France in 1979 under the brand name Athymil, and soon thereafter in the UK as Norval. Investigators conducting clinical trials in the US submitted fraudulent data, and it was never approved in the US.[45]: 21 [46]: 318
Mianserin was one of the first antidepressants to reach the UK market that was less dangerous than the tricyclic antidepressants in overdose; as of 2012 it was not prescribed much in the UK.[47]
Society and culture

Generic names
Mianserin is the English and German generic name of the drug and its INN and BAN, while mianserin hydrochloride is its USAN, BANM, and JAN. Its generic name in French and its DCF are miansérine, in Spanish and Italian and its DCIT are mianserina, and in Latin is mianserinum.[48][1][49][2]
Brand names
Mianserin is marketed in many countries mainly under the brand name Tolvon. It is also available throughout the world under a variety of other brand names including Athymil, Bonserin, Bolvidon, Deprevon, Lantanon, Lerivon, Lumin, Miansan, Serelan, Tetramide, and Tolvin among others.[1][2][48]
Availability
Mianserin is not approved for use in the United States, but is available in the United Kingdom and other European countries.[50][51] A mianserin generic drug received TGA approval in May 1996 and is available in Australia.[52]
Research
The use of mianserin to help people with schizophrenia who are being treated with antipsychotics has been studied in clinical trials; the outcome is unclear.[53][54]
References
- ^ a b c Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 689–. ISBN 978-3-88763-075-1.
- ^ a b c "International brands for mianserin". Drugs.com. Retrieved 20 August 2017.
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ a b c d e f g h i j Truven Health Analytics, Inc. Drugdex System (Internet) [cited 2013 Sep 29]. Greenwood Village, CO: Thomsen Healthcare; 2013.
- ^ a b c d e "Tolvon Product Information" (PDF). Medicines. AU: Merck Sharp & Dohme. Archived from the original (PDF) on 2016-04-02. Retrieved 2013-10-05 – via GuildLink.
- ^ a b c d e f g "Mianserin 30 mg film-coated tablets". UK Electronic Medicines Compendium. January 2014. Retrieved 20 August 2017.
- ^ "A Comparison of the Physicochemical and Biological Properties of Mirtazapine and Mianserin". Journal of pharmacy and pharmacology. Oxford University Press. April 2011. Retrieved 29 January 2022.
- ^ "Que faire devant une insomnie" [What to do when facing insomnia]. Sommeil (in French). Lyon, FR: University of Lyon.
- ^ "Traitement des troubles du sommeil" [Treatment of the troubles of sleep]. Research gate (in French). Archived from the original on 2019-03-30.
- ^ Kuniyoshi M, Arikawa K, Miura C, Inanaga K (June 1989). "Panic anxiety after abrupt discontinuation of mianserin". The Japanese Journal of Psychiatry and Neurology. 43 (2): 155–59. doi:10.1111/j.1440-1819.1989.tb02564.x. PMID 2796025. S2CID 527031.
- ^ Taylor D, Paton C, Kapur S, Taylor D (2012). The Maudsley Prescribing Guidelines in Psychiatry (11th ed.). Chichester, West Sussex: John Wiley & Sons.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- ^ a b c Tatsumi M, Groshan K, Blakely RD, Richelson E (December 1997). "Pharmacological profile of antidepressants and related compounds at human monoamine transporters". European Journal of Pharmacology. 340 (2–3): 249–258. doi:10.1016/s0014-2999(97)01393-9. PMID 9537821.
- ^ a b Boess FG, Martin IL (1994). "Molecular biology of 5-HT receptors". Neuropharmacology. 33 (3–4): 275–317. doi:10.1016/0028-3908(94)90059-0. PMID 7984267. S2CID 35553281.
- ^ a b c Toll L, Berzetei-Gurske IP, Polgar WE, Brandt SR, Adapa ID, Rodriguez L, et al. (March 1998). "Standard binding and functional assays related to medications development division testing for potential cocaine and opiate narcotic treatment medications". NIDA Research Monograph. 178: 440–466. PMID 9686407.
- ^ Matsumoto I, Combs MR, Jones DJ (February 1992). "Characterization of 5-hydroxytryptamine1B receptors in rat spinal cord via [125I]iodocyanopindolol binding and inhibition of [3H]-5-hydroxytryptamine release". The Journal of Pharmacology and Experimental Therapeutics. 260 (2): 614–626. PMID 1738111.
- ^ Peroutka SJ, Switzer JA, Hamik A (1989). "Identification of 5-hydroxytryptamine1D binding sites in human brain membranes". Synapse. 3 (1): 61–66. doi:10.1002/syn.890030109. PMID 2521959. S2CID 23503235.
- ^ Waeber C, Schoeffter P, Palacios JM, Hoyer D (June 1988). "Molecular pharmacology of 5-HT1D recognition sites: radioligand binding studies in human, pig and calf brain membranes". Naunyn-Schmiedeberg's Archives of Pharmacology. 337 (6): 595–601. doi:10.1007/bf00175783. PMID 2975354. S2CID 21344978.
- ^ a b Millan MJ, Newman-Tancredi A, Audinot V, Cussac D, Lejeune F, Nicolas JP, et al. (February 2000). "Agonist and antagonist actions of yohimbine as compared to fluparoxan at alpha(2)-adrenergic receptors (AR)s, serotonin (5-HT)(1A), 5-HT(1B), 5-HT(1D) and dopamine D(2) and D(3) receptors. Significance for the modulation of frontocortical monoaminergic transmission and depressive states". Synapse. 35 (2): 79–95. doi:10.1002/(SICI)1098-2396(200002)35:2<79::AID-SYN1>3.0.CO;2-X. PMID 10611634. S2CID 20221398.
- ^ Elliott JM, Kent A (July 1989). "Comparison of [125I]iodolysergic acid diethylamide binding in human frontal cortex and platelet tissue". Journal of Neurochemistry. 53 (1): 191–196. doi:10.1111/j.1471-4159.1989.tb07313.x. PMID 2723656. S2CID 25820829.
- ^ Bonhaus DW, Flippin LA, Greenhouse RJ, Jaime S, Rocha C, Dawson M, et al. (July 1999). "RS-127445: a selective, high affinity, orally bioavailable 5-HT2B receptor antagonist". British Journal of Pharmacology. 127 (5): 1075–1082. doi:10.1038/sj.bjp.0702632. PMC 1566110. PMID 10455251.
- ^ Bonhaus DW, Bach C, DeSouza A, Salazar FH, Matsuoka BD, Zuppan P, et al. (June 1995). "The pharmacology and distribution of human 5-hydroxytryptamine2B (5-HT2B) receptor gene products: comparison with 5-HT2A and 5-HT2C receptors". British Journal of Pharmacology. 115 (4): 622–628. doi:10.1111/j.1476-5381.1995.tb14977.x. PMC 1908489. PMID 7582481.
- ^ Wainscott DB, Lucaites VL, Kursar JD, Baez M, Nelson DL (February 1996). "Pharmacologic characterization of the human 5-hydroxytryptamine2B receptor: evidence for species differences". The Journal of Pharmacology and Experimental Therapeutics. 276 (2): 720–727. PMID 8632342.
- ^ Nelson DR, Thomas DR (May 1989). "[3H]-BRL 43694 (Granisetron), a specific ligand for 5-HT3 binding sites in rat brain cortical membranes". Biochemical Pharmacology. 38 (10): 1693–1695. doi:10.1016/0006-2952(89)90319-5. PMID 2543418.
- ^ Kohen R, Metcalf MA, Khan N, Druck T, Huebner K, Lachowicz JE, et al. (January 1996). "Cloning, characterization, and chromosomal localization of a human 5-HT6 serotonin receptor". Journal of Neurochemistry. 66 (1): 47–56. doi:10.1046/j.1471-4159.1996.66010047.x. PMID 8522988. S2CID 35874409.
- ^ Hirst WD, Abrahamsen B, Blaney FE, Calver AR, Aloj L, Price GW, Medhurst AD (December 2003). "Differences in the central nervous system distribution and pharmacology of the mouse 5-hydroxytryptamine-6 receptor compared with rat and human receptors investigated by radioligand binding, site-directed mutagenesis, and molecular modeling". Molecular Pharmacology. 64 (6): 1295–1308. doi:10.1124/mol.64.6.1295. PMID 14645659.
- ^ a b c d e Fernández J, Alonso JM, Andrés JI, Cid JM, Díaz A, Iturrino L, et al. (March 2005). "Discovery of new tetracyclic tetrahydrofuran derivatives as potential broad-spectrum psychotropic agents". Journal of Medicinal Chemistry. 48 (6): 1709–1712. doi:10.1021/jm049632c. PMID 15771415.
- ^ Jasper JR, Kosaka A, To ZP, Chang DJ, Eglen RM (September 1997). "Cloning, expression and pharmacology of a truncated splice variant of the human 5-HT7 receptor (h5-HT7b)". British Journal of Pharmacology. 122 (1): 126–132. doi:10.1038/sj.bjp.0701336. PMC 1564895. PMID 9298538.
- ^ Eglen RM, Jasper JR, Chang DJ, Martin GR (April 1997). "The 5-HT7 receptor: orphan found". Trends in Pharmacological Sciences. 18 (4): 104–107. doi:10.1016/s0165-6147(97)01043-2. PMID 9149537.
- ^ a b c d e f g Richelson E, Nelson A (July 1984). "Antagonism by antidepressants of neurotransmitter receptors of normal human brain in vitro". The Journal of Pharmacology and Experimental Therapeutics. 230 (1): 94–102. PMID 6086881.
- ^ Weinshank RL, Zgombick JM, Macchi M, Adham N, Lichtblau H, Branchek TA, Hartig PR (November 1990). "Cloning, expression, and pharmacological characterization of a human alpha 2B-adrenergic receptor". Molecular Pharmacology. 38 (5): 681–688. PMID 2172775.
- ^ Grandy DK, Marchionni MA, Makam H, Stofko RE, Alfano M, Frothingham L, et al. (December 1989). "Cloning of the cDNA and gene for a human D2 dopamine receptor". Proceedings of the National Academy of Sciences of the United States of America. 86 (24): 9762–9766. Bibcode:1989PNAS...86.9762G. doi:10.1073/pnas.86.24.9762. PMC 298581. PMID 2532362.
- ^ Ghoneim OM, Legere JA, Golbraikh A, Tropsha A, Booth RG (October 2006). "Novel ligands for the human histamine H1 receptor: synthesis, pharmacology, and comparative molecular field analysis studies of 2-dimethylamino-5-(6)-phenyl-1,2,3,4-tetrahydronaphthalenes". Bioorganic & Medicinal Chemistry. 14 (19): 6640–6658. doi:10.1016/j.bmc.2006.05.077. PMID 16782354.
- ^ a b c Appl H, Holzammer T, Dove S, Haen E, Strasser A, Seifert R (February 2012). "Interactions of recombinant human histamine H₁R, H₂R, H₃R, and H₄R receptors with 34 antidepressants and antipsychotics". Naunyn-Schmiedeberg's Archives of Pharmacology. 385 (2): 145–170. doi:10.1007/s00210-011-0704-0. PMID 22033803. S2CID 14274150.
- ^ Nguyen T, Shapiro DA, George SR, Setola V, Lee DK, Cheng R, et al. (March 2001). "Discovery of a novel member of the histamine receptor family". Molecular Pharmacology. 59 (3): 427–433. doi:10.1124/mol.59.3.427. PMID 11179435.
- ^ a b c d Olianas MC, Dedoni S, Onali P (November 2012). "The atypical antidepressant mianserin exhibits agonist activity at κ-opioid receptors". British Journal of Pharmacology. 167 (6): 1329–1341. doi:10.1111/j.1476-5381.2012.02078.x. PMC 3504997. PMID 22708686.
- ^ Leonard B, Richelson H (2000). "Synaptic Effects of Antidepressants: Relationship to Their Therapeutic and Adverse Effects". In Buckley JL, Waddington PF (eds.). Schizophrenia and Mood Disorders: The New Drug Therapies in Clinical Practice. Oxford: Butterworth-Heinemann. pp. 67–84. ISBN 978-0-7506-4096-1.
- ^ Müller G (8 May 2006). "Target Family-directed Masterkeys in Chemogenomics". In Kubinyi H, Müller G, Mannhold R, Folkers G (eds.). Chemogenomics in Drug Discovery: A Medicinal Chemistry Perspective. John Wiley & Sons. p. 25. ISBN 978-3-527-60402-9.
- ^ Onali P, Dedoni S, Olianas MC (January 2010). "Direct agonist activity of tricyclic antidepressants at distinct opioid receptor subtypes". The Journal of Pharmacology and Experimental Therapeutics. 332 (1): 255–265. doi:10.1124/jpet.109.159939. PMID 19828880. S2CID 18893305.
- ^ Dwivedi Y, Agrawal AK, Rizavi HS, Pandey GN (December 2002). "Antidepressants reduce phosphoinositide-specific phospholipase C (PI-PLC) activity and the mRNA and protein expression of selective PLC beta 1 isozyme in rat brain". Neuropharmacology. 43 (8): 1269–1279. doi:10.1016/S0028-3908(02)00253-8. PMID 12527476. S2CID 22105260.
- ^ Kishi T, Iwata N (February 2014). "Meta-analysis of noradrenergic and specific serotonergic antidepressant use in schizophrenia". The International Journal of Neuropsychopharmacology. 17 (2): 343–354. doi:10.1017/S1461145713000667. PMID 23823741.
- ^ Gillman PK (March 2006). "A systematic review of the serotonergic effects of mirtazapine in humans: implications for its dual action status". Human Psychopharmacology. 21 (2): 117–125. doi:10.1002/hup.750. PMID 16342227. S2CID 23442056.
- ^ "Mirtazapine label – Australia". GuildLink, Pharmacy Guild of Australia. 27 May 2016. Archived from the original on 21 November 2018. Retrieved 21 June 2017.
- ^ Kelder J, Funke C, De Boer T, Delbressine L, Leysen D, Nickolson V (April 1997). "A comparison of the physicochemical and biological properties of mirtazapine and mianserin". The Journal of Pharmacy and Pharmacology. 49 (4): 403–11. doi:10.1111/j.2042-7158.1997.tb06814.x. PMID 9232538. S2CID 12270528.
- ^ Shorter E (2005). A historical dictionary of psychiatry. Oxford: Oxford Univ. Press. ISBN 978-0-19-517668-1.
- ^ Stahl SM (2013). Stahl's essential psychopharmacology : neuroscientific basis and practical application (4th ed.). Cambridge: Cambridge University Press. ISBN 978-1-10702598-1.
- ^ Pratt JP (2012). "29. Affective Disorders". In Walker R, Whittlesea C (eds.). Clinical pharmacy and therapeutics (5th ed.). Edinburgh: Churchill Livingston/Elsevier. p. 472. ISBN 978-0-70204293-5.
- ^ a b Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 822–. ISBN 978-1-4757-2085-3.
- ^ Morton IK, Hall JM (31 October 1999). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 181–. ISBN 978-0-7514-0499-9.
- ^ Gelenberg AJ, Schoonover SC (29 June 2013). "Major Psychiatric Disorders: Depression". In Gelenberg AJ, Bassuk EL, Schoonover SC (eds.). The Practitioner's Guide to Psychoactive Drugs. Springer Science & Business Media. pp. 39–. ISBN 978-1-4757-1137-0.
- ^ Quitkin FM, Taylor BP (24 May 2013). "Antidepressants". In Klein DF, Rowland LP (eds.). Current Psychotherapeutic Drugs. Routledge. pp. 57–. ISBN 978-1-135-06284-2.
- ^ "Lumin Mianserin hydrochloride product information" (PDF). Medicines. AlphaPharm – via GuildLink.
- ^ Terevnikov V, Joffe G, Stenberg JH (May 2015). "Randomized Controlled Trials of Add-On Antidepressants in Schizophrenia". The International Journal of Neuropsychopharmacology. 18 (9): pyv049. doi:10.1093/ijnp/pyv049. PMC 4576515. PMID 25991654.
- ^ Vernon JA, Grudnikoff E, Seidman AJ, Frazier TW, Vemulapalli MS, Pareek P, et al. (November 2014). "Antidepressants for cognitive impairment in schizophrenia--a systematic review and meta-analysis". Schizophrenia Research. 159 (2–3): 385–394. doi:10.1016/j.schres.2014.08.015. PMC 4252251. PMID 25240772.
Further reading
- Peet M, Behagel H (1978). "Mianserin: a decade of scientific development". British Journal of Clinical Pharmacology. 5 (Suppl 1): 5S–9S. doi:10.1111/j.1365-2125.1978.tb04567.x. PMC 1429213. PMID 623702.

















