| The development of data science: Implications for education, employment, research, and the data revolution for sustainable development 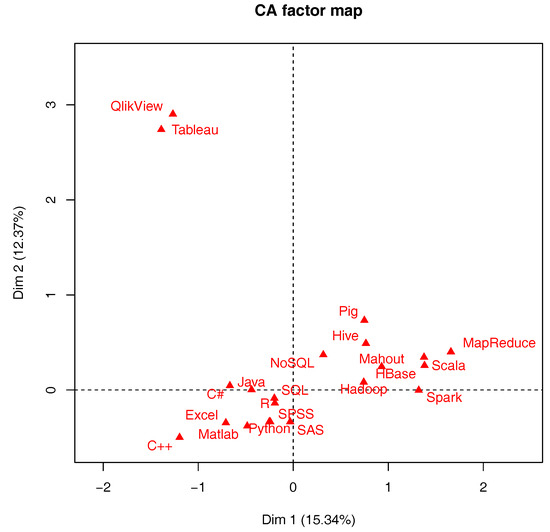 In this 2018 paper by Murtagh and Devlin, a historical and professional perspective on data science and how collaborative work across multiple disciplines is increasingly common to data science. This "convergence and bridging of disciplines" strengthens methodology transfer and collaborative effort, and the integration of data and analytics guides approaches to data management. But education, research, and application challenges still await data scientists. The takeaway for the authors is that "the importance is noted of how data science builds collaboratively on other domains, potentially with innovative methodologies and practice," In this 2018 paper by Murtagh and Devlin, a historical and professional perspective on data science and how collaborative work across multiple disciplines is increasingly common to data science. This "convergence and bridging of disciplines" strengthens methodology transfer and collaborative effort, and the integration of data and analytics guides approaches to data management. But education, research, and application challenges still await data scientists. The takeaway for the authors is that "the importance is noted of how data science builds collaboratively on other domains, potentially with innovative methodologies and practice,"
GeoFIS: An open-source decision support tool for precision agriculture data 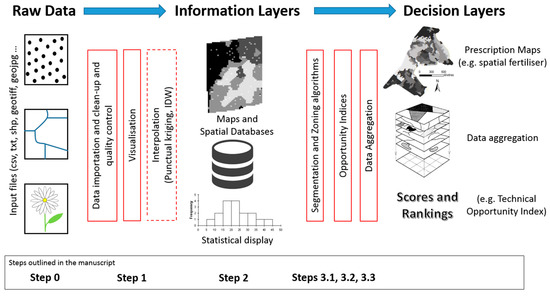 Scientists everywhere nod to the power of big data but are still left to develop tools to manage it. This is just as true in the field of agriculture, where practitioners of precision agriculture are still developing tools to do their work better. Leroux et al. have been developing their own solution, GeoFIS, to better handle geolocalized data visualization and analysis. In this 2018 paper, they use three case studies to show off how GeoFIS visualizes data, processes it, and incorporates associated data (metadata and industry knowledge) for improved agricultural outcomes. They conclude that the software fills a significant gap while also promoting the adoption of precision agriculture practices. Scientists everywhere nod to the power of big data but are still left to develop tools to manage it. This is just as true in the field of agriculture, where practitioners of precision agriculture are still developing tools to do their work better. Leroux et al. have been developing their own solution, GeoFIS, to better handle geolocalized data visualization and analysis. In this 2018 paper, they use three case studies to show off how GeoFIS visualizes data, processes it, and incorporates associated data (metadata and industry knowledge) for improved agricultural outcomes. They conclude that the software fills a significant gap while also promoting the adoption of precision agriculture practices. |