Type a search term to find related articles by LIMS subject matter experts gathered from the most trusted and dynamic collaboration tools in the laboratory informatics industry.
 | |
| Clinical data | |
|---|---|
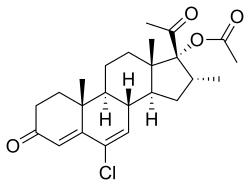
| Other names | Clomagestone acetate; SH-741; 6-Chloro-17α-acetoxy-16α-methylpregna-4,6-diene-3,20-dione |
| Drug class | Progestogen; Progestogen ester |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C24H31ClO4 |
| Molar mass | 418.96 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Clomegestone acetate (USAN) (developmental code name SH-741), or clomagestone acetate, also known as 6-chloro-17α-acetoxy-16α-methylpregna-4,6-diene-3,20-dione, is a steroidal progestin of the 17α-hydroxyprogesterone group which was developed as an oral contraceptive but was never marketed.[1][2] It is the acetate ester of clomegestone, which, similarly to clomegestone acetate, was never marketed.[1] Clomegestone acetate is also the 17-desoxy cogener of clometherone, and is somewhat more potent in comparison.[3] Similarly to cyproterone acetate, clomegestone acetate has been found to alter insulin receptor concentrations in adipose tissue,[4] and this may indicate the presence of glucocorticoid activity.[5][6]